Mixtures
Complete the sentence.
Liquid is matter with a definite ________ but no definite ________ .
Liquid is matter with a definite volume but no definite shape .
What is the formula for density?
Density = Mass / volume
What group and period is Iron located?
Group 8 and Period 4
Which are the two types of pure substances?
A) elements and solutions
B) matter and mixtures
C) elements and compounds
D) homogeneous and heterogeneous mixtures
C) elements and compounds
What is a precipitate?
A solid forms after two liquids are mixed.
Which statement accurately compares the arrangement of atoms in the image to their arrangement in solids?
A.) Atoms in the image are close together and move freely, while atoms in solids are far apart and move freely.
B.) Atoms in the image are close together and vibrate in place, while atoms in solids are close together and move freely.
C.) Atoms in the image are far apart and move freely, while atoms in solids are close together and vibrate in place.
C.) Atoms in the image are far apart and move freely, while atoms in solids are close together and vibrate in place.
It is easier to swim in the salt water of the ocean than in a fresh water of a lake.
What statement explains this phenomena?
A) Salt water is less dense than fresh water, so your body will float more easily in sea water.
B) Salt water is more dense than fresh water, so your body will float more easily in sea water.
C) Waves in the ocean make it easier to swim.
D) Your body is less dense in sea water than in fresh water so you float easier.
B) Salt water is more dense than fresh water, so your body will float more easily in sea water.
A scientist is working with an element that is a gas at room temperature and does not react with other elements. Which characteristic BEST describes this element?
A.) Metal
B.) Member of Group 18
C.) Metalloid
D.) Member of Group 2
B.) Member of Group 18
What type of matter is shown in the photo?
Heterogeneous mixture
Which observations are an example of a physical change rather than a chemical change?

C, E, F
Identify the state of matter for each substance.
A - Liquid
B - Gas
C- Solid

A, B, C
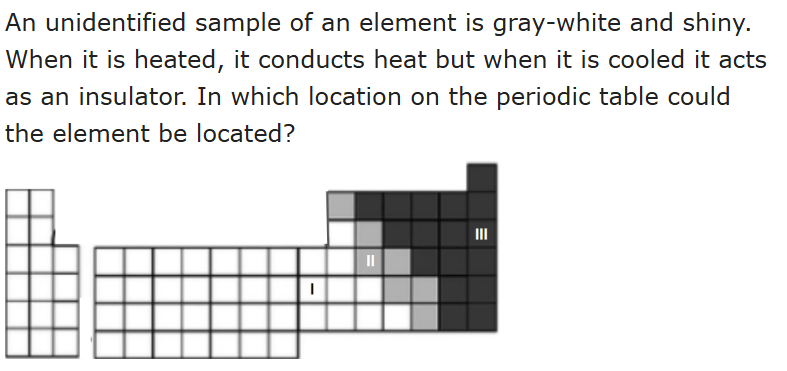
ll only (metalloids)
You have a jar filled with a clear liquid. You can't see anything suspended in the liquid. However, when you taste it, you notice a bitter flavor. Based on this information, the liquid is most likely-
-Heterogeneous
-Homogeneous
-Pure Substance
homogeneous mixture
Which is not a clue that could indicate a chemical change?
A.) Change in color
B.) Change in shape
C.) Change in energy
D.) Change in odor
B.) Change in shape
List the states of matter in order from least to greatest of the kinetic energy of the atoms and molecules.
Solid, Liquid, Gas
A rock has a mass of 30g and a volume of 2cm3. What is the density of the rock?
15 g/cm3
Identify element 1 and element 2.
Element 1 - nonmetal
Element 2 - metal
Which statement is NOT true?
A) A pure substance can be separated into elements and compounds by physical methods.
B) A pure substance is made up of only one type of particles.
C) Each substance has unique properties, such as the melting point and boiling point.
D) Elements and compounds are pure substances.
A) A pure substance can be separated into elements and compounds by physical methods.
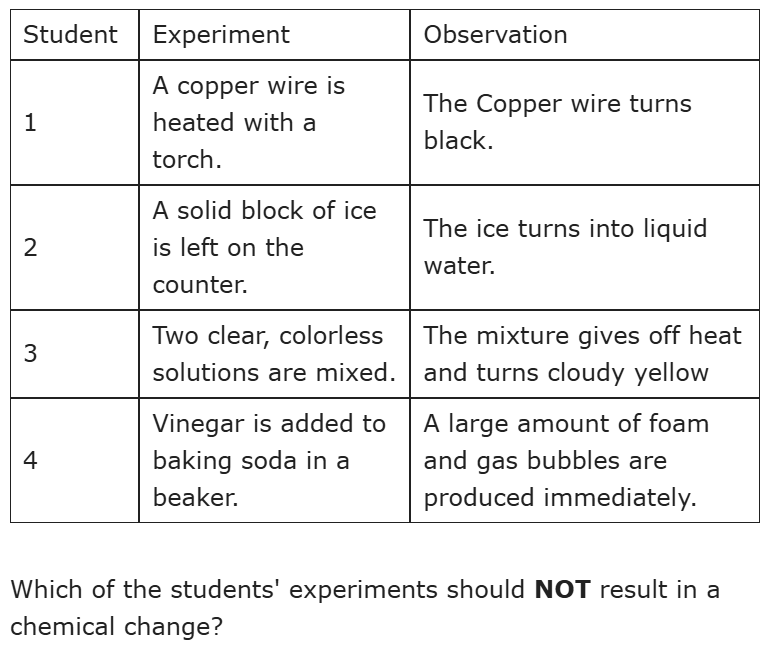
Student 2
Kinetic energy increases
List the substances in order from least dense to most dense.

A, D, B, C
Elements that share common characteristics are grouped together on the periodic table. In which part of the table would you MOST LIKELY find an element that is a gas at room temperature?
On the RIGHT (Nonmetals)
Which statement about the mixtures is NOT correct?
A) The substances of a mixture preserve their identity and individual properties.
B) All mixtures are solutions.
C) Mixtures can be separated through physical changes, though some mixtures are hard to separate.
D) Magnets, sieves, filters, and other materials can be used to separate mixtures.
B) All mixtures are solutions.
A scientist mixes two clear liquids, Liquid X and Liquid Y, in a beaker. After mixing, the beaker feels noticeably warmer to the touch. The resulting liquid remains clear, and no bubbles are observed.
Which evidence from the experiment BEST suggests that a new substance has been formed?
The temperature of the beaker increased.