Helium
What is He?
elements you need to create Air
What is Nitrogen and Oxygen?
The name of horizontal rows on the periodic table
What is periods?
The amount of atoms in this bohr model
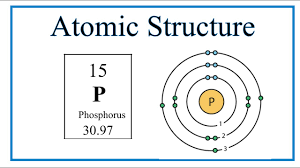
What is 15?
Atom definition
What is the basic unit of a chemical element?
Potassium
What is K?
The most common type of homogeneous mixture
What is a solid,liquid or gas?
The vertical rows on the periodic table
What is groups or families?
This is what the lewis dot diagram shows
What is how the valence electrons are distributed around the atoms in a molecule
Molecule definition
What is the smallest particle of a substance?
Zirconium
What is Zr?
Elements you need to create gunpowder
What is sulfur, potassium and carbon?
This is what the periodic table is sorted by
What is the increasing mass of each atom?
This is what each dot represents in a lewis dot structure
What is a electron?
Atom examples
What is Neon, Hydrogen, Iron, and calcium?
Mercury
What is Hg?
Elements to create seawater
This is what the number on the top left is called on a element block
What is atomic number?
when the bohr model was invented
what is 1913?
Molecule examples
What is, water, Nitrogen, Ozone, Calcium Oxide, and glucose?
Lead
What is Pb?
elements you need to create crude oil
What is organic compounds, mainly hydrocarbons?
This is what the number on the bottom is called on an element block
What is the atomic weight?
These are the main points of the bohr model
What is electrons orbit the nucleus in orbits that have a set size and energy?
Is table salt a molecule or atom
what is molecule?