Lithium, commonly used in car and cell phone batteries, is found in nature as Li-6 and Li-7, with the latter being far more abundant on Earth. Both isotopes have this many protons and this many neutrons.
What is 3 protons in both and 3 neutrons in Li-6 and 4 in Li-7?
This is the reason the periodic table is called the periodic table.
What is because chemical characteristics repeat themselves over and over again?
This is the oxidation number of alkali metals AND this is the reason why these elements are found in this oxidation state.
What is +1, and because alkali Earth metals want to have a full valence shell (8 electrons) so they give away a negative electron to become stable.
What is fluorine, Ken?
The molar mass of dihydrogen monoxide.
What is 18 grams/mole
This is the location, mass and charge of protons, neutrons and electrons in the atom.
What is:
Protons(+1) and neutrons(no charge) are in the nucleus, each with a mass of 1 amu. Electrons (negative charge) are outside the nucleus and have no mass. 
This is the halogen in the 4th period.
What is bromine?
Adding silver nitrate to sodium chloride to result in this white cloudy substance. What is the name of the product in general and what is the name of the compound?
![]()
![]()
What is a precipitate?
The formula for rubidium phosphide.
What is Rb3P?
Hydrogen peroxide, or H2O2, is used to disinfect wounds. Is this the empirical formula, molecular formula or both? Why?
What is the molecular formula, Alex? It has a name and we can assume it is found in nature like this (and that it is the MF)
and you can reduce it to HO so it is NOT the EF.
 Chex mix is this type of mixture.
Chex mix is this type of mixture.
What a heterogeneous mixture?
This is the electron configuration of manganese (#25)?
What is 1s22s22p63s23p64s23d5?
Compound A has a melting point of 801 degrees C, is a cubic crystalline shape, and conducts electricity when dissolved in water.
Compound B has a melting point of 86 degrees C, is an amorphic solid and does not conduct electricity when dissolved in water.
This compound represents a bond that is held together by a sharing of electrons.
What is compound B?
The name of Co3(PO4)2.
What is cobalt II phosphate?
If you mix a strip of magnesium in an aqueous solution of copper II sulfate, will a reaction take place?
Yes, Alex.
Copper has a melting point of 1083 degrees C, a density of 8.92 g/cm^3 and a specific heat of 0.385 J/g C. These describe ____________properties of copper.
What are physical properties, Ken?
An excited electron goes from n= 4 to n=2. What is the wavelength of light emitted (don't forget units)? Does this wave have more or less energy than n=4 to n=3?
434 nanometers; it has more energy than 1875 nanometers emitted from an excited electron going from n=4 to n=3.
This is a correct identification of ionic, polar and nonpolar covalent compounds
H2O, PbO, Br2
What is:
H2O= polar covalent,
PbO = ionic, and
Br2= nonpolar covalent
Which has higher electronegitivity and ionization energy, lithium or oxygen?
What is oxygen, Ken? (They both increase up and to the right on the PT.)
Indium (III) oxide, is used in manufacturing touch screens for smart phones and tablets. Indium is relatively rare and the US geological Survey says we will run out of extractable indium in less than 20 years. Calculate the percent of indium found in In2O3
What is 82.8 percent, Alex?
This is the atomic number and the mass number of the most common isotope of fluorine (#9). AND this identifies the significance of these numbers.
Atomic number = 9 (number of protons)
Mass number= 19 amus (9 protons and 10 neutrons)
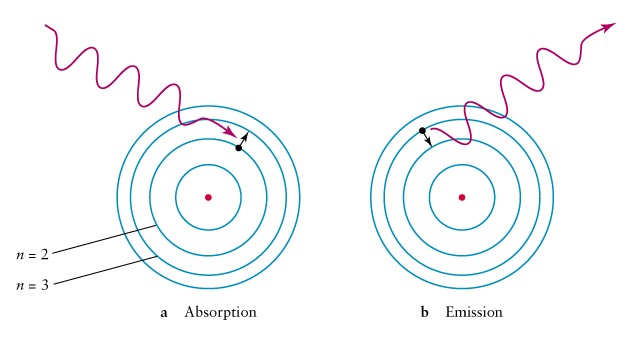
This is a good explanation of what happens and why when an electron goes from ground to excited back to ground state.
What is the electron closer to the nucleus (ground state) absorbs energy, jumps away (excited state) and returns to ground releasing a photon of light, with a certain wavelength and frequency.
The name of the group that has the following electron dot diagram:
What are the chalcogens, Ken?
What is the name of S2O4?
What is disulfur tetraoxide and manganese IV hydroxide?
These are the number of molecules in 30 grams of water.
What is 1 x 1024 molecules?