This law states that energy can neither be created nor destroyed
the law of conservation of energy
The state of matter with the most amount of energy.
Gass
Increasing _________ will increase my gravitational potential energy.
Height
Heat transfers from ____________ to ___________
Hot to cold
Does mass affect temperature?
No
The sum of potential and kinetic energy
mechanical energy
An object with a higher temperature has particles that are moving this way compared to a cooler object.
They move Faster
If I want more kinetic energy I can increase my __________ or ____________
Mass or speed
This is what moves between two things that are different tempertures
Heat
What is an example of a good thermal conductor
Metals (copper, steel, aluminum......)
Material that slows down the transfer of heat.
Insulator
What is conduction
Heat transfer from direct contact
When energy changes from one form to another, it is called an ____________When energy moves from one object to another, it is called an ____________.
transformation, transfer
Air currents heat up and cool off due to this type of heat transfer.
Convection
Grabbing a hot frying pan is an example of what heat transfer
conduction
A measure of the average kinetic energy of all particles in an object or substance. Temperature does not depend on the material or the type of particles in a substance
Temperature
What is this an example of?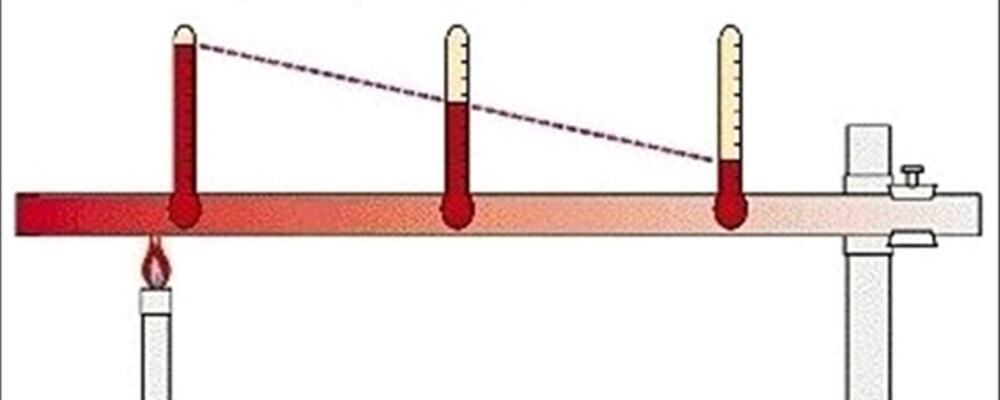
The breaking of bonds when you digest food is an example of _______ energy
chemical
Heat will continue to transfer until __________ is reached.
Thermal equilibrium
Thermal energy is affected by state of matter, ___________, and ____________
mass and temperature
The measurement of the total amount of kinetic energy of all the particles in an object or substance
Thermal energy
What one has more thermal energy 
the iglue
Name the Energy Transformations in a TV Remote
Mechanical, Chemical, Electrical, Radiant
A microwave uses what type of heat transfer?
Radiation
When the molecules begin to slow down and move closer together what is happening to the thermal energy of that substance?
It is decreasing its thermal energy