This is a testable explanation or "educated guess" for a scientific observation
These are the three subatomic particles found in an atom.
What are protons, neutrons, and electrons?
This is a substance made of two or more different elements chemically bonded together, such as H2O.
What is a compound?
Describe the law of conservation of matter/mass.
What is matter cannot be created or destroyed, it just changes from?
This type of property can be observed without changing the substance into something new
What is a physical property?
What is the independent variable?
Dependent?
What is the variable that is changed by the scientist, what is the variable that is measured or observed?
This is where protons and neutrons are located.
What is the nucleus?
Phosphorus and Nitrogen are examples of these "pure substances" that cannot be broken down by chemical means
what is an element?
Draw the energy of a gas
What is high energy, particles moving all around?
This physical property describes how much "stuff" is in a given amount of space (D= M/V)
What is density?
Data that involves numbers and measurements is called this
What is quantitative?
Most of an atom's mass is concentrated in this central region
What is the nucleus?
KoolAid, Salt water, and Air are examples of this substance
What is a mixture?
What is a solid?
This property involves a chemical reaction and completely changes the substance
what is a chemical reaction?
Decide the independent and dependent variables for the below scenario
The respiration rate of a goldfish is measured. The goldfish is then placed in cold water and the respiration rate is measured again.
IV: water temperature
DV: respiration rate
Has matter been conserved?
2CH4 + O2 -> 2CO2 + 3H2O
What is matter is not conserved?
Toxicity and flammability are examples of this type of property, which describes how a substance reacts with others
What is a chemical property?
Decide the independent and dependent variables for the below scenario
Bacteria are grown in a petri dish. One side of the dish is sprayed with an antibiotic. After a week, the number of bacteria colonies are counted on each side.
IV: Antibiotic
DV: Number of bacteria colonies
Identify the element below.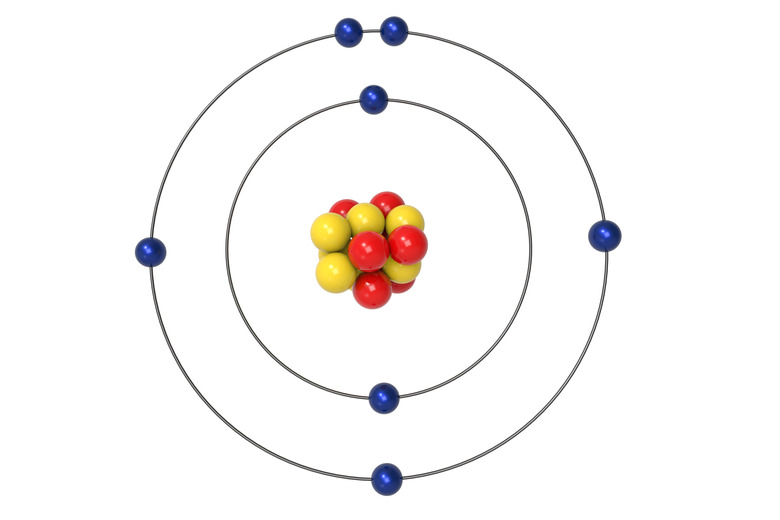
What is nitrogen?
Describe the difference between compounds and mixtures.
What is compounds are chemically bonded and mixtures are not?
Has matter been conserved?
Zn + 2HCl-> ZnCl2 +H2
What is matter is conserved?
On the pH scale, a substance with a rating of 3 (like acid rain) is considered this.
What is acid?