A technique that measures the energy required to remove electrons from atoms in the gas phase
What is photoelectron spectroscopy (PES)?
An instrument used to determine atomic mass based on mass-to-charge ratio
What is a mass spectrometer?
The difference between valence electrons and electrons assigned in a Lewis structure
What is formal charge?
The internuclear distance where attraction balances repulsion
What is bond length?
Molecular geometry of SO2
What is bent or angular?
Weak IMF present in all molecules
What are London dispersion forces?
Ratio used to convert between substances
What is a mole ratio?
Relationship between rate and concentration
What is the rate law?
The x-axis quantity on a PES spectrum that increases from right to left
What is binding energy?
The quantity represented on the y-axis of a mass spectrum
What is relative abundance?
A structure minimizing formal charges with negative charge on the most electronegative atom
What is the most stable Lewis structure?
Relationship between bond order and bond length
What is higher bond order gives shorter bond length?
Electron geometry with four electron domains
What is tetrahedral?
IMF responsible for water's high boiling point
What is hydrogen bonding?
Reactant consumed first
What is the limiting reactant?
Units of k for a first-order reaction
What is s-1?
This feature of a PES peak corresponds to the relative number of electrons in a sublevel
What does peak area (or height) represent?
The reason chlorine shows two major peaks at m/z ≈ 35 and 37
What is the existence of Cl-35 and Cl-37 isotopes?
The correct formal charge on nitrogen in NH4+
What is +1?
Depth of potential energy well indicates this property
What is bond energy?
The correct number of resonance structures of ozone: O3
What is 2?
Strongest IMF in methanol
What is hydrogen bonding?
Maximum predicted product mass
What is theoretical yield?
Linear 1/[A] vs time plot indicates this order
What is second order?
The peak that would appear at the highest binding energy for sodium
What is the 1s electron peak?
The calculated value that combines isotopic mass and relative abundance
What is weighted average atomic mass?
The justification for drawing multiple valid Lewis structures for nitrate
What is resonance due to electron delocalization?
The bond length of Na-Cl according to the green line on this graph
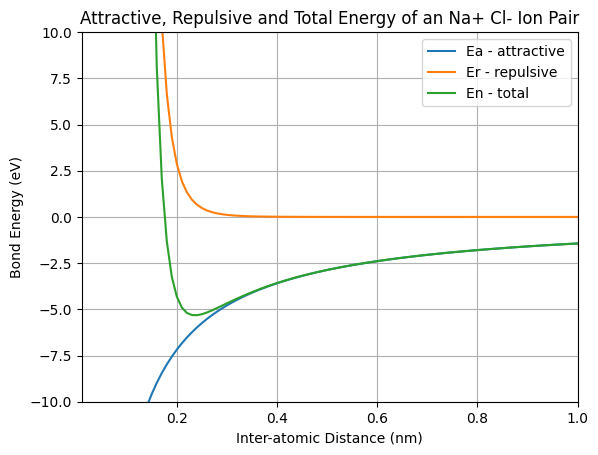
What is ~0.22 nm?
The bond angles in this molecule
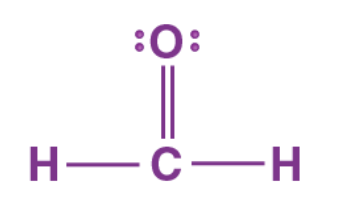
What is 120o?
Property directly affected by IMF strength
What is boiling point? or similar
Formula comparing actual to theoretical yield
What is percent yield?
Why rate laws cannot come from coefficients
What is rate law must be determined experimentally?
Or What is not all reactions are elementary?
The PES for this element looks like this.
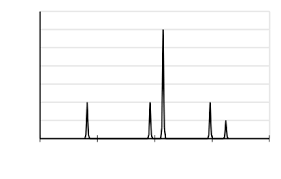
What is aluminum?
The mass spectrum of this element looks like this
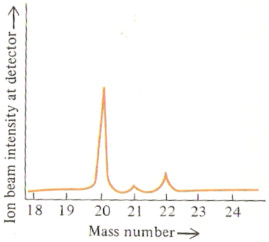
What is neon?
Why resonance does not imply oscillation between structures
What is a resonance hybrid representing an average structure?
Why breaking a chemical bond requires energy
What is overcoming electrostatic attraction?
This is the most realistic bond order for a carbonate ion
CO32-
What is 4/3 or 1.33?
The one expected to have the weakest IMF of these two

What is methanol?
Mass of CO2 produced from combustion of 10.0g C2H6
What is approximately 29.3 g CO2?
The order for a reaction with a constant rate, rate = k
What is zeroth order?