What theory predicts the shape of a molecule based on the idea that pairs of electrons will orient themselves to be as far apart as possible?
Valence Shell Electron Pair Repulsion Theory (VSEPR)
According to the electronegativity values table, a bond is classified as Nonpolar Covalent if the difference in electronegativity is less than or equal to what number?
0.4
Why do Ionic compounds not use prefixes in their naming?
they have 1 ratio when balancing the charges of the cation and the anion
What do we use in naming covalent compounds to indicate the number of each atom in the molecule?
We use Greek prefixes like mono-, di-, tri, etc...
What types of elements for ionic compounds and covalent compounds
Ionic: metal + nonmetal
Covalent: nonmetal + nonmetal
What specific shape name describes a molecule with a central atom bonded to three other atoms with one unshared (lone) pair of electrons, such as in the molecule NH3?
Pyramidal
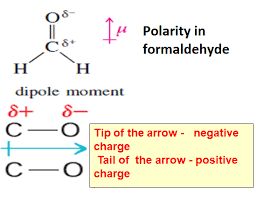
The arrow must always face towards which specific element in the bond when drawing dipole moments on a molecule?
The one with the higher Electronegativity
To balance the 2+ charge of the magnesium cation and the - charge of the chloride anion, how would you write the chemical formula?
MgCl2
What are the two other naming rules besides greek prefixes when naming covalent compounds?
1. the second elements ending changes to -ide
2. when the first element contains only one atom we do not include the mono- prefix, but we do when the second element has only 1 atom
What is the difference in the way ionic compounds and covalent compounds form bonds?
Covalent compounds share electrons and ionic compounds bond through a transfer of electrons from the cation to the anion
What are the 5 specific shapes we discussed within the VSEPR Theory?
Linear, Bent, Pyramidal, Trigonal Planar, Tetrahedral
If its VSEPR shape has this property, a molecule is often considered nonpolar, even if it contains polar covalent bonds.
If it is symmetrical
Why do some chemical names contain roman numerals?
Some transition metals make multiple ions and the roman numeral shows the charge that the cation has taken
What is the chemical formula for dihydrogen monoxide?
H2O
Which type of compound has a higher melting point and why?
Think about the following
-intermolecular forces vs. intramolecular force
Ionic compounds have a stronger intramolecular forces which forms a rigid highly organized crystal lattice. lattice is a repeated pattern of attraction between cations and anions. The strong electrostatic attraction holds them tightly together. While some covalent compounds have intermolecular forces, this only occurs when there are polar charges in a molecule. This attraction is not as strong as an ionic bond. It takes more energy to separate the strong forces in ionic compounds, resulting in a high melting point.
What specific style of drawing uses solid and dashed lines to show bonds coming towards or going away from the viewer and is often required alongside Lewis structures to depict 3D geometry?
Dash-Wedge Drawing
If the difference in electronegativity between two atoms is greater than 1.8, the bond is considered to be this type.
Ionic
Why do some chemical formulas contain parenthesis?
They go around a polyatomic ion when the molecule contains more than one group of those bonded atoms
What is the name for the following chemical formula: PCl5
Phosphorus Pentachloride
Unlike covalent compounds, ionic compounds have the ability to do this when dissolved in water, making them electrolytes.
They can conduct electricity, but not when solid
Draw the Lewis Dot structure and VSEPR shape for CH2O
Add any partial charges, dipole moments, and net dipole

Is CH3F a nonpolar or polar molecule?
Polar
Name the following chemical compound; PbO2
Lead (IV) Oxide
Why do we need to indicate the number of atoms in the compound? Why can we not just list the two elements?
Covalent compounds bond in various ratios. They can form single, double and triple bonds between the same two elements
Using both picture and words explain why ionic compounds are brittle.
