This part of an atom has a positive charge.
proton
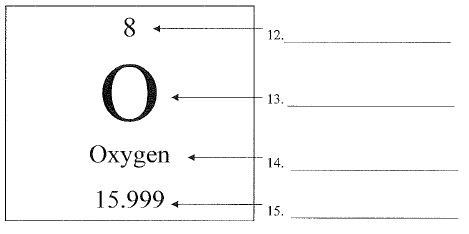
What is number 15 called?
atomic mass
H2 +O2 --> H2O
Potassium has this many valence electrons.
1
NH4 has this many atoms.
5
-->
yields
_C + _O2 --> _CO2
1C + 1O2 --> 1CO2
This is an atom that has the same number of protons, neutrons and electrons. It has no charge.
neutral atom
Protons and neutrons are found in this center part of the atom.
nucleus
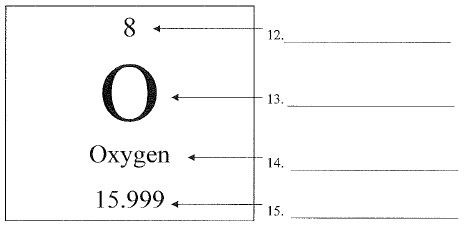
What is number 14 called?
element name or chemical name
Cl2 + NaBr --> NaCl + Br2
single replacement
Scandium has this many valence electrons.
3
2C2H3N5 has this many atoms.
20
What is the 4 in this formula?
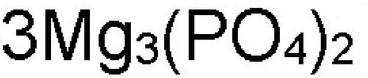
subscript
_Al2O3 --> _Al + _O2
2Al2O3 --> 4Al + 3O2
This is an atom that has gained neutrons and now has a higher atomic mass than a neutral atom.
heavy isotope
The part of the atom that has no charge (neutral).
neutron
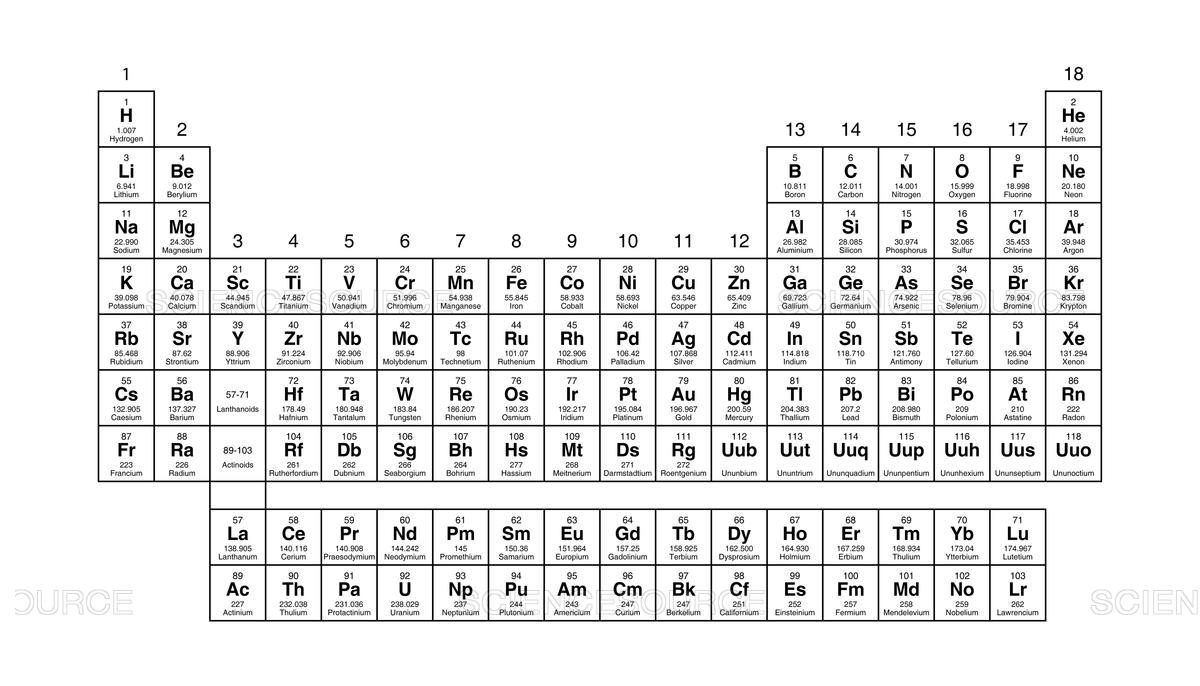 The columns on the Periodic Table are called this.
The columns on the Periodic Table are called this.
groups
CaCO3 --> CaO + CO2
decomposition
Arsenic has this many valence electrons.
15
2C4H8FCOOH has this many HYDROGEN atoms.
18
What is the 3 in the following formula?
3H2O
coefficient
_Mg + _HCl --> _MgCl2 + _H2
1Mg + 2HCl --> 1MgCl2 + 1H2
This is an atom that has gained electrons. It now has a negative charge.
anion
These negative parts of the atom are found in levels orbiting around the center.
electrons
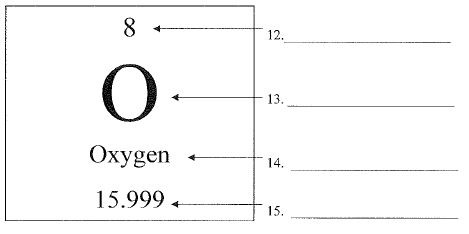
What is number 12 called?
Atomic Number
Na2S + HCl --> NaCl + H2S
double replacement
Xenon has this many valence electrons.
0 or 18
2S4H5(F2Ti3)3 would have this many atoms.
48
What is Mg in this formula?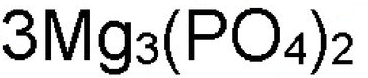
chemical symbol or element symbol
_CH4 + _O2 --> _CO2 + _H2O
1CH4 + 2O2 --> 1CO2 + 2H2O
This is an atom that has LOST neutrons and now has a smaller atomic mass than a neutral atom.
light isotope
List how many electrons can fit into all the shells (energy levels) in order from the first shell (level) to the seventh.
2, 8, 8, 18, 18, 32, 32
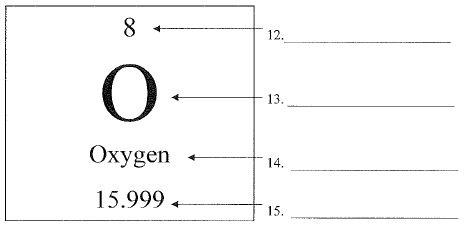
What is number 13 called?
chemical symbol or elemental symbol
CH4 + O2 --> CO2 + H2O
combustion
Boron has this many valence electrons.
3
4(NH4)3PO4 would have this many atoms.
80
What is between the 4 and the 2 in this formula?
3Ba3(PO4)2
parenthesis or brackets
_NH4OH --> _H2O + _NH3
1NH4OH --> 1H2O + 1NH3
This is an atom that has LOST electrons and now has more protons than electrons. It has a positive charge.
cation
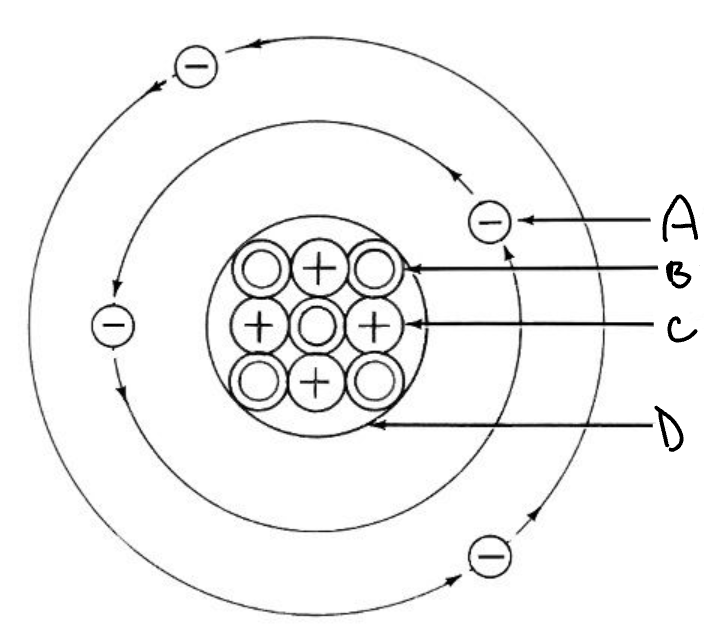 Name all 4 things that the letters are pointing to.
Name all 4 things that the letters are pointing to.
A-electron
B-neutron
C-proton
D-nucleus
You find the number of neutrons in an atom by doing this.
atomic mass - atomic number
K2CrO4 + AgNO3 --> Ag2CrO4 + KNO3
Double Replacement
Aluminum has this many valence electrons.
3
3Cr(NH3)6(NO3)3 would have this many NITROGEN atoms.
27
What does a coefficient do?
multiplies everything in the formula or tells you how many molecules there are
_CaF2 + _AlN --> _Ca3N2 + _AlF3
3CaF2 + 2AlN --> 1Ca3N2 + 2AlF3
What do we call an atom that does not have equal protons and electrons and has a charge?
ion
Protons, neutrons and electrons are all types of what?
Subatomic particles
 The rows on the Periodic Table are called this.
The rows on the Periodic Table are called this.
periods
H2CO3 --> H2O + CO2
decomposition
List all of the "Noble Numbers"
2, 10, 18, 36, 54, 86
2K4Fe(SCN)6Cr2(SO4)3 has how many total atoms?
80
What does a subscript do?
Tells you how many atoms you have of the element symbol that came before it
_C6H14 + _O2 --> _CO2 + _H2O
2C6H14 + 19O2 -->12CO2 + 14H2O
What do we call an atom that has more or less neutrons than it is supposed to have which changes its atomic mass?
isotope
Who created the Periodic Table?
Dmitri Mendeleev
(NH4)3PO4 + Pb(NO3)4 --> Pb(PO4)4 + NH4NO3
double replacement
Name one of the two most active groups on the Periodic Table.
Alkali Metals or Halogens
4(NH4)3PO4 + 3Pb(NO3)4 --> 1Pb(PO4)4 + 12NH4NO3
262
Connects a smaller set inside a formula. They always come in pairs.
_(NH4)3PO4 + _Pb(NO3)4 --> _Pb(PO4)4 + _NH4NO3
4(NH4)3PO4 + 3Pb(NO3)4 --> 1Pb3(PO4)4 + 12NH4NO3