Which below are an example of a chemical change?
A) The rusting of a penny
B) Water freezing into ice
C) Water vapor condensing into water
D) Stone breaking apartA) The rusting of a penny
Which below is an example of a physical change?
A) Cooking chicken.
B) Burning a log.
C) Forming a precipitate.
D) Chopping carrots.
D) Chopping carrots.
A type of change where the physical properties of matter change.
Physical Change
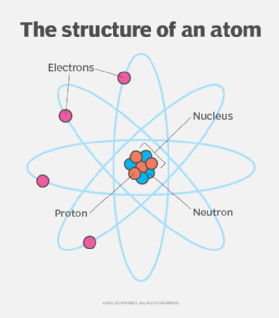
Positively charged particle that forms the nucleus and determines atomic number.
Protons
The negatively charged particle that orbits the nucleus of an atom.
Electron
A group of atoms bonded together, representing the smallest fundamental unit of a chemical compound that can take part in a chemical reaction.
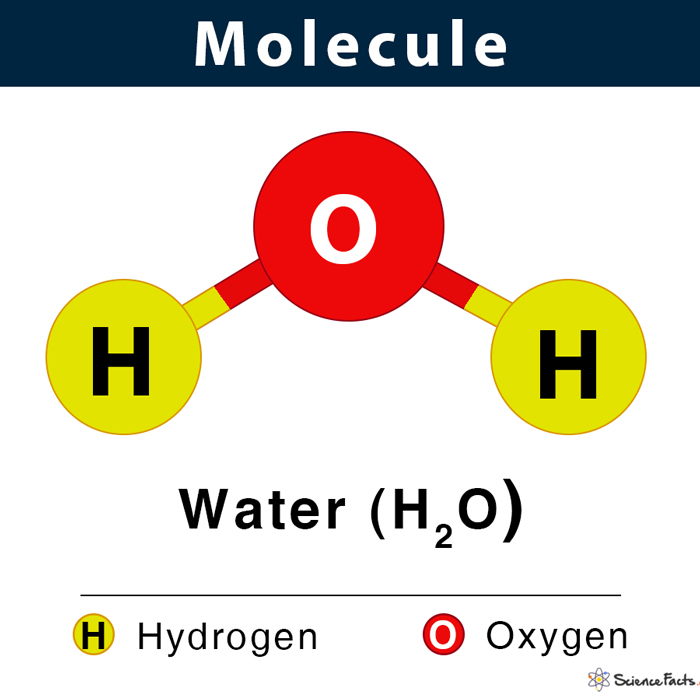
Molecule
A physical or conceptual representation of a system of ideas, events or processes.
A model
A mixture where throughout the solution the composition is not uniform.
Heterogeneous Mixture
John weighs 12 gram of ice in a cup. Afterwards he then melts the ice that was in the cup into water. John then re-weighs the cup. How much will it weigh?
12 grams
Compound, Element, or Mixture.

Mixture
A mixture in which the composition is uniform throughout the mixture.
Homogenous Mixture
The basic unit of a chemical element.
Atom
Compound, Element, or Mixture

Compound
In a chemical reaction mass is neither created nor destroyed.
Law of Conservation of Mass
Compound, Element, or Mixture

Element
A process in which one or more substances, also called reactants, are converted to one or more different substances, known as products. Requires energy in order to occur.
Chemical Reaction or Chemical Change
Energy is neither created or destoryed, it only changes form.
Law of Conservation of Energy
The minor component in a solution, dissolved in the solvent.
Solute
A substance with a pH=7 is...
Neutral
A Substance with a pH<7 is...
Acidic
pH stands for...
Potential Hydrogen
A substance with a pH>7 is...
Alkaline
The area where electrons are found around the nucleus.
Electron Cloud
Solvent
On the graph below what change in physical property is occurring where the arrows are indicating?

Phase Change
Atomic Theory has developed over time via.....
A series of discoveries supported by scientific evidence and thorough experimentation.
This subatomic particle has no charge
Neutron
Paula was testing the fountain water from her school. Paula found that when using a strip of pH testing paper that it would turn red with a pH of 5.4. Is the water acidic, alkaline, or neutral
Acidic
Which element below is Copper(Cu) Most chemically similar to.
 A) (Au) Silver
A) (Au) Silver
B) (Br) Bromine
C) (O) Oxygen
D) (Ca) Calcium
A) Silver
This atomic model was proposed by...

J.J. Thompson