Ionic bonding occurs between what 2 types of elements?
metals and non-metals
Covalent bonding occurs between what 2 types of elements?
non-metals and non-metals
What 3 letters do ionic compounds end with?
-IDE
What 3 letters do covalent compounds usually end with?
-IDE
This type of element makes up polyatomic ions.
What are nonmetals
Which electrons are involved in ionic bonding?
valence electrons
Which electrons are involved in covalent bonding?
valence electrons
Name the following compound: SrCl2
What is strontium chloride
What is the prefix for 4 atoms?
tetra-
How are polyatomic ionic compounds different from Typ I and Type II ionic compounds?
What is polyatomic ionic compounds contain 3 or more different elements.
calcium ion symbol
Ca2+
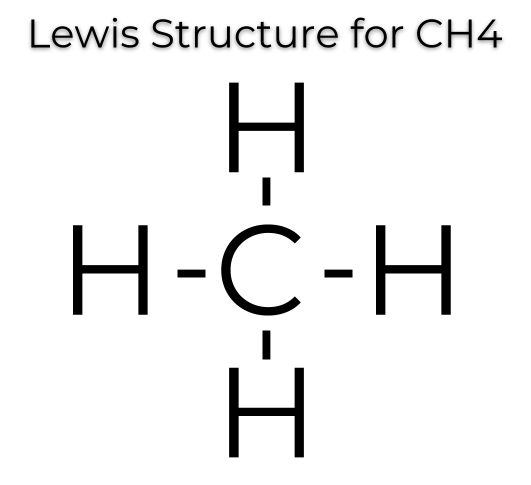
What is the Lewis Structure for CH4?
CH4, carbon tetrahydride
TRUE OR FALSE: The name of CaCl2 is calcium dichloride.
FALSE. The real name is calcium chloride.
Name the following compound: N2O2
dinitrogen dioxide
Name the compound: Li(NO3)
lithium nitrate
Electrons are doing what in ionic bonding?
Electrons are transferred from metal atoms to nonmetals atoms.
What is a chemical formula? (you can give an example)
a formula that gives you the atoms and the number of atoms for each element
Ex. H2O or CaCl2
Name the following compound: HgO
mercury (II) oxide
Name the following compound: P2O4
diphosphorus tetroxide
Chemical formula for potassium hydroxide
KOH
What type of ionic compound has only 2 different atoms bonded together?
binary ionic compound
Electrons are doing what in covalent bonding?
"sharing" electrons between them (we know they are really fighting over them)
What is the name of CuCl2?
copper (II) chloride
what is the chemical formula for tetraphosphorus decoxide
P4O10
chemical formula for potassium phosphate
K3(PO4)