This law states that the change in internal energy equals heat added plus work done
First Law (Energy Conservation)
What happens to a system’s internal energy when it releases heat and no work is done?
The system’s internal energy decreases.
What is the device that is used in chemistry to insulate a chemical reaction called?
Is the formation of chemical Bonds Endothermic or Exothermic?
Exothermic
Draw the structure for pentane
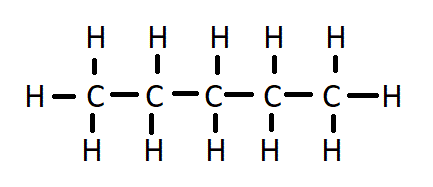
This quantity must increase for any spontaneous process in an isolated system
Entropy (Second Law)
A system absorbs 150 J of heat and does 40 J of work on the surroundings.
What is the change in internal energy?
ΔU = +110 J
During heating, why might a substance with a high specific heat show a slower temperature increase even with continuous heat input?
A substance with a high specific heat requires more energy to raise its temperature
Is water evaporating endothermic or exothermic?
Endothermic: energy is being absorbed by the water
Draw this organic compound: C4H8

This law implies that reaching this temperature is impossible in a finite number of steps
Absolute Zero (Third Law)
If ΔU = +120 J and the system does 50 J of work, how much heat was added to the system?
q = +170 J
170 J of heat was added to the system.
A 250 g sample of an unknown material requires 3000 J of heat to increase its temperature by 15°C.
Is this substance is more similar to water or a metal?
A Metal
I thi Reaction Endothermic or Exothermic?

Exothermic
Calculate the bond enthalpy of this reaction

6494 - 8528 = ~-2034 kJ/mol
This law is what allows thermometers to measure temperature accurately
Zeroth Law (Thermoequilibrium)
Heat of −250 J flows out of a system, and the internal energy decreases by 400 J. What is the work done?
w = −150 J
If a 500 g piece of copper (0.39J/g°C) experiences a temperature change of 30°C, what is the heat transfer (Q)?
5850 J
A reaction absorbs 250 J of heat but releases 100 J through work. Is the overall process endothermic or exothermic? Explain in terms of q.
Endothermic, because the system absorbs 250 J of heat (positive q)
Calculate the bond enthalpy for the reaction.
Is the reaction endothermic or exothermic?
678 - 862 = ~ -184 kJ/mol
Exothermic
A system undergoes a spontaneous process where entropy decreases locally but increases overall, this law still holds because of this condition
The Second Law (the total entropy of the universe increases)
A system undergoes the following three processes:
Step 1: Absorbs 350 J of heat and does 120 J of work
Step 2: Releases 200 J of heat while 80 J of work is done on the system
Step 3: Does 150 J of work with no heat transfer
What is the total change in internal energy (ΔU) for the entire process?
ΔU= 230−120−150= −40 J
ΔU= −40 J
A 100 g piece of iron at 90°C is placed in 200 g of water at 20°C. The specific heat of iron is 0.45 J/g°C. What is the final temperature of the system?
23.5°C
Draw the potential energy diagram (graph) for an exothermic reaction, label Enthalpy (ΔH) and Activation energy (Ea)

Calculate the enthalpy of combustion of ethane using bond enthalpies
2C₂H₆ + 7O₂ → 4CO₂ + 6H₂O
4573 - 5994 = ~ -1421 kJ/mol