The tiny pieces that all matter is made of.
Atoms
What was the brown substance in the water in Westfield?
Rust
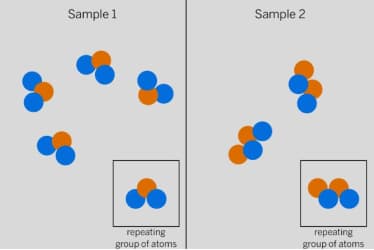
These substances will have __________ properties.
different
The temperature at which a substance changes from the solid phase to the liquid phase is
Melting point
Is the following chemical reaction balanced?
H2O + NH4 -----> NH2 + H2
No. The products are missing an O and 2H's
An object, diagram, or computer program that helps us understand something by making it simpler or easier to see.
Model
Can rust be created without fertilizer?
No
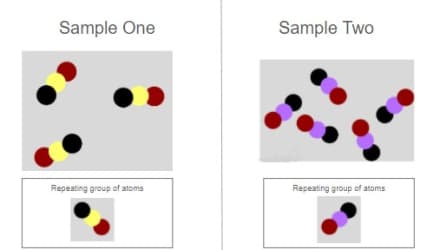
These substances have the exact same properties. True or False?
False
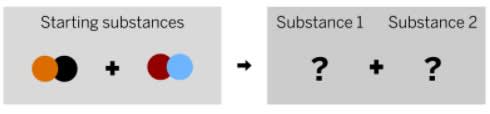
What is a possible product these reactants can form?
red, orange
blue, black
red, black
blue, orange
During a chemical reaction, atoms cannot be ______ or ______.
Created or destroyed
A process in which atoms rearrange to form new substances
Chemical Reaction
What two substances created rust?
During a chemical reaction, ______ of the atoms that make up the reactants rearrange to form the products.
All
A beginning substance or substances in a chemical reaction
Reactant

 devorah is a food chemist, and she is developing a chemical to help foods stay fresh longer. She mixed a red liquid substance and a colorless solid substance together in a sealed container. The diagram above shows the repeating groups of atoms that make up the two starting substances.
devorah is a food chemist, and she is developing a chemical to help foods stay fresh longer. She mixed a red liquid substance and a colorless solid substance together in a sealed container. The diagram above shows the repeating groups of atoms that make up the two starting substances.
After mixing, Devorah found two white substances in the sealed container. (Nothing had escaped.) Which of the diagrams to the left shows the repeating groups of atoms that make up the ending substances?
C
An ending substance that is made during a chemical reaction.
Product
What other substance was found in Westfield's water?
Sodium nitrite (NaNO2) or
Potassium chromate (K2CrO4)
Sodium nitrite (NaNO2)
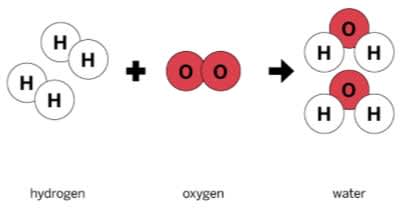
In the reaction shown, hydrogen and oxygen are the _______.
Reactants
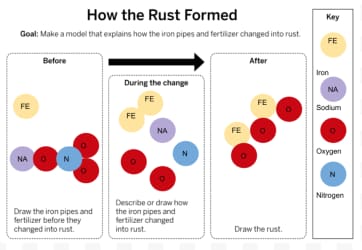
Can substances change into different substances?
yes, because atoms can be rearranged to form different substances
Is the following chemical reaction balanced?
O2 + H2O + CO2 -----> H2 + O2 + O3
No. It is missing the C.
Something that can be observed about a substance, such as color, smell or boiling point.
Property
What was discovered in Westfield's well?
Fertilizer
In this chemical equation, which is one of the products? 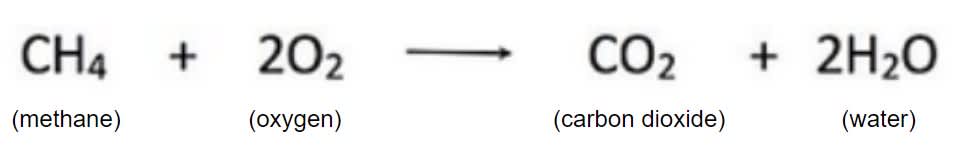
Water
Carbon Dioxide
What happens to the atoms of the reactants that create different substances in the products?
*to change the order or position of something
rearrange
Is the following chemical reaction balanced?
NH4 + CO2 -------> H2O + H2O + N + C
Yes.
