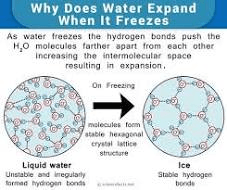
Why is frozen water (ice) less dense allowing it to float?
These are molecules have the same chemical formula but different structures
What are Isomers
The number of bonds that carbon can form.
What is 4
What functional group makes up water?
Hydroxyl (-OH)
Define pH and describe the pH scale. What is considered an acid, neutral and basic/alkaline.
What is the power of hydrogen or measure of the concentration of H+ ions in a solutions. Scale is 1-14. PH values less than 7 are acid, 7 is neutral and greater than 7 are basic/alkaline.
This is the name of the substance dissolved in a solvent.
What is Solute
What is the difference between organic and inorganic molecules?
Organic molecules contain carbon and inorganic molecules do not contain carbon

Describe the type of reaction that involves breaking a polymer down into monomers?
What is hydrolysis - addition of water to break bonds
This functional group is written as -C=O
What is carbonyl group
This is the name to describe the cohesive forces between water molecules that allows some insects to walk on water.
What is Surface Tension
What is the name given for water being attracted to other water molecules. What is the name for water being attracted to other molecules/substances?
What is cohesion? What is adhesion?
What are the 6 essential elements for life? These elements form the major biological molecules, nucleic acids, proteins, carbohydrates, and lipids.
What is carbon, hydrogen, oxygen, nitrogen, phosphorus and sulfur.
What elements are found in proteins?
C, H, O, N, S
This is the "amino" functional group
What is -NH2
A biomolecule's function is dictated by its _________.
What is structure
This is the heat required to change the temp of 1 gram of a substance 1 degree C.
What is Specific Heat? High specific heat = less fluctuation in water temperature
What type of bond is found between Oxygen and Hydrogen in a water molecule?
covalent
What are the monomers for all 4 biomolecules?
Carbohydrates - monosaccharides
Lipids - fatty acid & glycerol
Proteins - amino acids
Nucleic Acid - nucleotide
What functional group has -CH3?
This action is due to the pressure of cohesion and adhesion of water which cause the liquid to move up a tube.
What is Capillary Action
What are 3 differences between hydrophilic and hydrophobic substances? Describe each, dissolvability in water, polar or nonpolar
What is
Hydrophilic = water loving, can be dissolved in water, polar & ionic
Hydrophobic = water fearing/hating, cannot be dissolved in water, nonpolar
_________ bonds result from the attractive force between a hydrogen atom and a very electronegative atom such as N and O. This type of bond exists between water molecules.
What is hydrogen bond
What biomolecule is this?
What is a nucleotide - nucleic acid biomolecule
Which two functional groups make up a protein?
What is carboxyl group - COOH and amino group - NH2
What year did Mrs. Irby graduate high school?
What is 2000