Solutions!
What is the predominant attractive force between the particles in MgF2?
Ionic
Which one of the following should have the lowest boiling point?
LiF, SiF4, PCl3, Cl2S, CH3COOH
SiF4
The heat of fusion of water is 6.01 kJ/mol. The heat capacity of liquid water is 75.3 J/mol*K. The conversion of 50.0 g of ice at 0.00 °C to liquid water at 0.00°C requires ________ kJ of heat.
16.7
An unsaturated solution is one that ________.

has a concentration lower than the solubility
What is the normal boiling point of this substance?
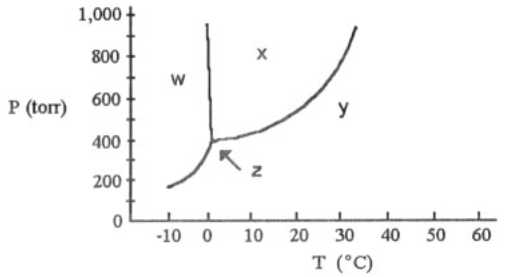
29
Hydrogen bonding is a special case of ________. [identify IMF]
dipole-dipole attractions
Which statements about viscosity are true?
(i) Viscosity increases as temperature decreases.
(ii) Viscosity increases as molecular weight increases
(iii) Viscosity increases as intermolecular forces increase.
All
Calculate the molality of a 15.3% (by mass) aqueous solution of hydrochloric acid
4.95 m
A sample of potassium nitrate (49.0 g) is dissolved in 100 g of water at 100 °C, with precautions taken to avoid evaporation of any water. The solution is cooled to 30.0 °C and no precipitate is observed. This solution is ________.

supersaturated
Which of the following substances is more likely to dissolve in CH3OH?
CCl4, CH3CH2OH, Kr, H2, N2
CH3CH2OH
Which one of the following exhibits dipole-dipole attraction between molecules?
Cl2, CF4, SnF3, SiS2, BI3
SnF3
Which is false?

The heat of sublimation is equal to the sum of the heat of vaporization and the heat of freezing.
Calculate the mole fraction of nitric acid of a 10.4% (by mass) aqueous solution of nitric acid.
3.21×10−2
Pairs of liquids that will mix in all proportions are called ________ liquids.
miscible
Pressure has an appreciable effect on the solubility of ________ in liquids.

Gases
Of the following substances, only ________ has London dispersion forces as its only intermolecular force.
NH3, CH4, CH3OH, H2S, HI
CH4
Vapor Pressure of a liquid

increases nonlinearly with increasing temperature
A solution is prepared by adding 30.00 g of lactose (milk sugar) to 110.0 g of water at 55 °C. The partial pressure of water above the solution is ________ torr. The vapor pressure of pure water at 55 °C is 118.0 torr. The MW of lactose is 342.3 g/mol.
116.3
The principal reason for the extremely low solubility of NaCl in benzene (C6H6) is the ________

weak solvation of Na+ and Cl- by C6H6
In a saturated solution of a salt in water, ________.

the rate of crystallization = the rate of dissolution
The dissolution of water in octane (C8H18) is prevented by ________.

hydrogen bonding between water molecules
Of the following, a 0.1 M aqueous solution of ________ will have the highest freezing point.

sucrose
On a clear day at sea level, with a temperature of 25°C, the partial pressure of N2 in air is 0.78 atm and the concentration of nitrogen in water is 5.3*10-4 M. When the partial pressure of N2 is ________ atm, the concentration in water is 4.3*10-3 M
6.3
The solubility of MnSO4 monohydrate in water at 20 °C is 70.0 g per 100.0 mL of water. A solution at 20 °C that is 0.401 M in MnSO4 monohydrate is best described as a(n) ________ solution. The formula weight of MnSO4 monohydrate is 168.97 g/mol.

unsaturated
A solution is prepared by dissolving 23.7 g of CaCl2 in 375 g of water. The density of the resulting solution is 1.05 g/mL. The concentration of Cl- in this solution is ________ M.
1.12