What is the definition of an isotope
same p diff neutrons
Orbital notation for Boron

What is the term for a charged atom
ion
Give the following info for the most abundant atom of Cesium (Cs) have?
mass number, atomic number, Protons, neutrons and electrons
MN: 133 AN: 55 P: 55 N: 78 E: 55
what is the term for a negative ion
anion
what is the term for a positive ion
cation
Bigger atomic radius: Fe or Ru?
Ru
Smaller electronegativity: Nb or Co?
electron conf. of argon
1s22s22p63s23p6
How many principle energy levels does an atom of Br have?
4
How many orbitals do the following sub levels have:
d and f
d: 5 f: 7
The ___________ accounts for almost all of the atom's mass. while the ____________ accounts for almost of the atom's volume
nucleus, electron cloud
Orbital diagram for Al
1s22s22p63s23p1
Write the protons, neutrons, electrons, atomic number and mass number for 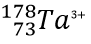
P: 73 N: 105 E: 70 AN: 73 MN: 178
Are these isotopes?
Atom Protons Neutrons Electrons
#1 7 7 10
#2 7 7 7
No ions
Put the following in order of atomic radius, 1-4 with 1 being the biggest and 4 being the smallest.
C, Rb, Li, K
1. Rb 2. K 3. Li 4. C
Isoptope 1: mass = 14.003074, 99.63%
Isotope 2: mass = 15.000108, 0.37%
What's average atomic mass
14.01
What is the atomic #, mass #, number of p, n, and e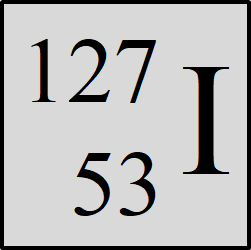
AN: 53 MN: 127 P and E: 53 N: 74
____________________ is the energy required to remove an electron
ionization energy
An element has an average mass of 74.922 amu. If there are only isotopes with a mass of 76 and a mass of 74 for this element, which isotope has the higher abundance?
74
An atom has 29 protons and 37 neutrons. What is the correct chemical symbol?
6629Cu
what element has the following electron configuration
1s22s22p63s23p64s23d104p65s24d9
Ag
Write the protons, neutrons, electrons, atomic number and mass number for
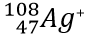
P: 47 N: 61 E: 46 AN: 47 MN: 108
How many protons, neutrons, and electrons does the following have
p: 25 n: 30 e: 18
What is a valence electron
electron in the outer most energy level
55% of a sample is found to have a mass of 28. The rest has a mass of 29. What is the average atomic mass of this sample?
28.45
Put the following in order of ionization energy, 1-4 with 1 being the most and 4 being the least.
Ca, K, P, S
What is the relationship between atomic radius and ionization energy? and Why?
A smaller atomic radius will have a larger ionization energy.
The most reactive elements are the ones who's electrons are the easiest to remove. Which metal in the 1st GROUP is the most reactive?
Francium
Draw the nuclear symbol for an element that has 34 protons and 36 electrons
7934Se2-