4 levels (1, 2, 3, 4)
Basic unit of matter and the smallest particle of an element is _______________
Atom
Atomic mass?

200.592
Atoms of the same element with same number of protons but different number of neutrons?
Isotope
Metric (SI) unit for measuring the amount of a SMALL substance
Mole (mol)
The area in an atom with a high chance of finding electrons is called
orbital
The outermost electrons are called
valence electrons
What is the name of the negatively-charged part of an atom?
Electron
# protons?

23
An unstable isotope that emits (releases) radiation until it decays into a stable isotope is called
Radioisotope (radioactive isotope) (radioactive)
To convert from moles to grams, you (add / multiply / divide / subtract)
Multiply
Which is a more stable state for electrons: ground or excited?
Ground
Which atomic theory said the atom is a positively-charged sphere with negatively-charged electrons throughout it?
Nuclear model
Name the 3 subatomic particles of an atom
Proton, neutron, electron
# electrons?
*Round to the nearest whole number

45
True or False: Isotopes of the same element have different masses.
True
# electrons?
*Round to the nearest whole number

24
Convert 1.5 moles of xenon to grams
*Round to 1 decimal place

196.9 grams
Which atomic theory became the basis for our understanding of atoms?
Nuclear model
What subatomic particle(s) is (are) found in the nucleus of an atom?
Protons, neutrons
# neutrons?
Round to the nearest full number

42
True or False:
The atomic mass of an element is a weighted average of all the element's isotopes.
True
# neutrons?
6
Convert 200 grams of NaCl to moles
*Round to 1 decimal place


3.4 moles
The first and most basic atomic theory was called
Atomism
Draw an atom and label the proton, neutron, and electron

# protons? # electrons? # neutrons?

p / e = 88
n = 138
How are these different?

Different masses OR # of neutrons
# protons? electrons? neutrons?

p = 10
e = 10
n = 10
Convert 2 moles methane (C2H8) to grams
*Round to 1 decimal place
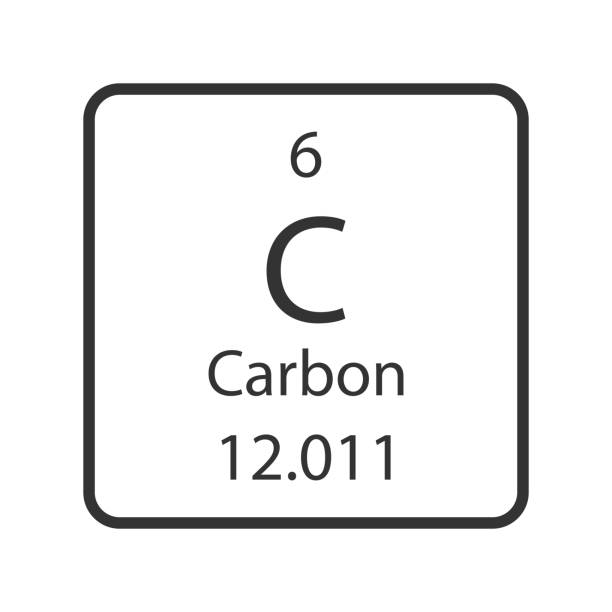
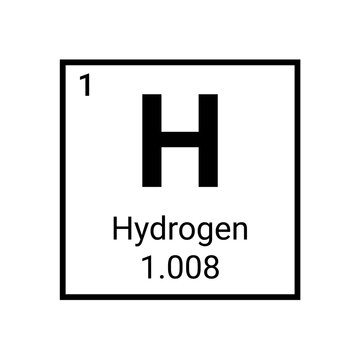
64.2 grams