Write 5 facts about electrons
negative
less mass than a proton or neutron
outside nucleus
288
outside shell electrons are called valence electrons
Which elements are the most reactive non-metals..why?
The atomic number refers to....
the number of protons
PIE
Color and label the Non-metals one color and color the metalloids another color. (leave the metals white)
hold your board up to check.... double check that Al was left white and H was colored the same color as the non-metals
Which atom is most reactive and why?

Chlorine because it has 7 valence electrons in its third energy level
Name an element that would have the same number of valence electrons as nitrogen and is also the same classification (metal, non-metal, metalloid).
Phosphorus
Color the group on the periodic table that would have elements that are shiny with 2 valence electrons
group 2/2a
Elements along the zig zag line are called________. Every element touching the line is included except___________.
Metalloids, aluminum
Where would you find an element that is a more malleable than sulfur but conducts less electricity than copper
metalloids
On your periodic table color in the section where you would find an element with the following characteristics:
2 valence electrons
metal
group 2/2a
Identify the Element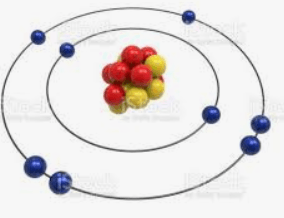
Oxygen
Which element is least reactive? Why?

Neon because is it has 8 valence electrons in the second energy level
Which subatomic particle is smaller than a proton?
electron
Name an element that react like Silicon but has a smaller mass.
Carbon
Elements in the same group have the same chemical properties because they have the same number of valence electrons.
What has to be true of every atom of Selenium?
34 Protons
How many valence electrons will an atom of Chlorine have
7
Describe the nucleus of an atom with 4 facts
How many Protons, Neutrons and Electrons are in an atom of Gold
P= 79, N=118, E= 79
Name the element and describe the reactivity
Potassium, Very reactive metal
___________ determines an elements ____________, and __________ ___________ determines an elements _c_________ p_________ like ___________.
Protons determine an elements identity and valence electrons determine chemical properties like reactivity.
Which pair of elements have the same number of atomic orbitals?
-Silver and Strontium
- Sodium and Strontium
-Silver and Sulfur
- Sulfur and Strontium
Silver and Strontium
Element Q is a very reactive metal and element Z has 3 less valence electrons. Element Z is a metalloid and both elements are in period 3. Identify element Z
Silicon
Write 5 facts about elements in group 18.
Noble gases, Chemically Stable, Unreactive, Inert, 8 valence electrons (except He has 2 VE), gases at room temperature
Mystery element "x" has 3 valence electrons and mystery element "y" has 2 more valence electrons than "X". If both elements are metals identify element "Y"
Bismuth