The number of protons in an atom's nucleus can be determined by looking at the ________ on the periodic table.
Atomic Number
The non-metals are located on this side of the periodic table?
Right side
What element has the symbol He?
Helium
What are the three subatomic particles?
Protons. Electrons. Neutrons.
How many electrons are in a neutral oxygen atom? (Hint: Oxygen has 8 protons)
8 electrons
Identify the charges of each of the subatomic particles.
protons +
neutrons 0
electrons -
This group is special because its outer most energy level is full.
Group 18 - Noble Gases
Identify the atomic number of the following element. What else does this number represent?
29; number of protons
What two particles are found IN the nucleus.
Protons and Neutrons
What is the name of the element with a mass number of 32 and 16 electrons?
Sulfur
The what particles compose the majority of the mass of an atom?
protons and neutrons
List two elements that are non-metals.
C, O, N, F, P, He, H, etc.
What is the mass number of this element? Identify the number of protons and neutrons in the element.

31; 15p; 16n
Where are the electrons located?
Electron Cloud
Name three types of transition metals.
Copper, gold, silver, nickel, titanium, etc
The rows of the periodic table are called ___________, and the columns of the periodic table are called _____.
Periods (rows)
Groups/Families (columns)
Name an element that is a metalloid. Then list one characteristic of metalloids.
Boron, Silicon, Antimony, Tellurium, Arsenic, Germanium
1. In between properties of metals/non metals
2. Semi-conductors
3. Solids at room temperature
Identify the number of protons, neutrons, and electrons in an atom of Be.
protons 4
neutrons 5
electrons 4
What atom is represented by the following Bohr diagram?

Fluorine
This element is located in Group 17 and has 3 energy levels.
Chlorine
What determines the number of energy levels an atom has?
Period Number
List three properties of metals.
1. High melting points
2. Good conductors of heat and electricity
3. Shiny solids at room temperature
4. Malleable
5. Ductile
Identify the number of neutrons in the given element. What is the overall net charge of this atom?
zero net charge
neutrons 0
Draw the Bohr diagram of Magnesium.
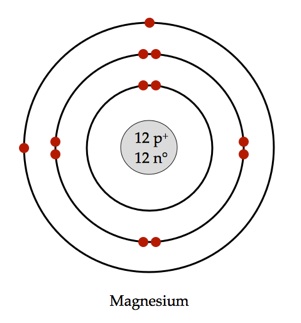
How many electrons can fit into each energy level?
Energy level 1:
Energy level 2:
Energy level 3:
2
8
8