What is the smallest unit of matter that can exist alone or in combination with others?
a. Element
b. Atom
c. Molecule
d. Nucleus
b. Atom
What is an element?
An element is a pure substance made of only ONE type of atom
What is a Molecule? Give an Example.
Two or more atoms bonded together.
Examples may vary.
How do you calculate the number of neutrons in an atom?
a. Same as the number of Protons
b. Atomic Number - Electrons
c. Atomic Number - Atomic Mass
d. Atomic Mass - Atomic Number
Atomic Mass - Atomic Number
What animal was hit by Mr. Schmid's car and then attacked him?
 Hawk
Hawk
What charges do protons, neutrons, and electrons each have?
Protons - positive (+)
Neutrons - neutral (0)
Electrons - negative (-)
What are the columns of the periodic table called?
What are the rows called?

Columns = groups / families
Rows = periods
This is a molecule of two hydrogen atoms (H2). What part of an atom determines how it bonds to others?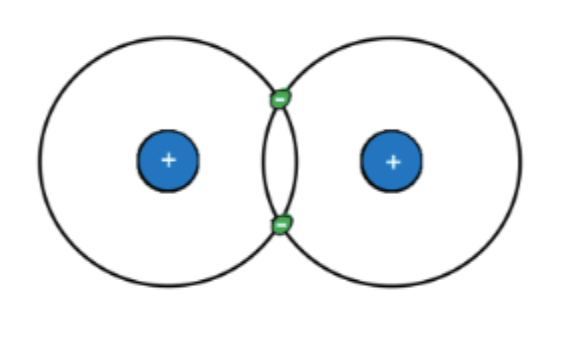
Electrons.
An atom's electrons (number, arrangment) determines how it will or won't bond with other atoms.
Potassium's Atomic Number is 19. Its Atomic Mass is 39.0983.
How many neutrons are in a Potassium Atom?

20 neutrons
What did Mr. Schmid Study in college?
a. Arabic and International Studies
b. Art and Biology
c. Physics and Chemistry
d. Music and Arabic
Where is the majority of an atom's mass?
Hint: this is where "atomic mass" comes from.
Nucleus (protons + neutrons)
In a given period (row) of the periodic table, what do all the atoms have in common?
a. Same properties
b. Same number of protons
c. Same number of electrons
d. Same number of electron shells
d. Same number of electron shells
This is a Carbon atom. How many more electrons does it need to have a full outer shell?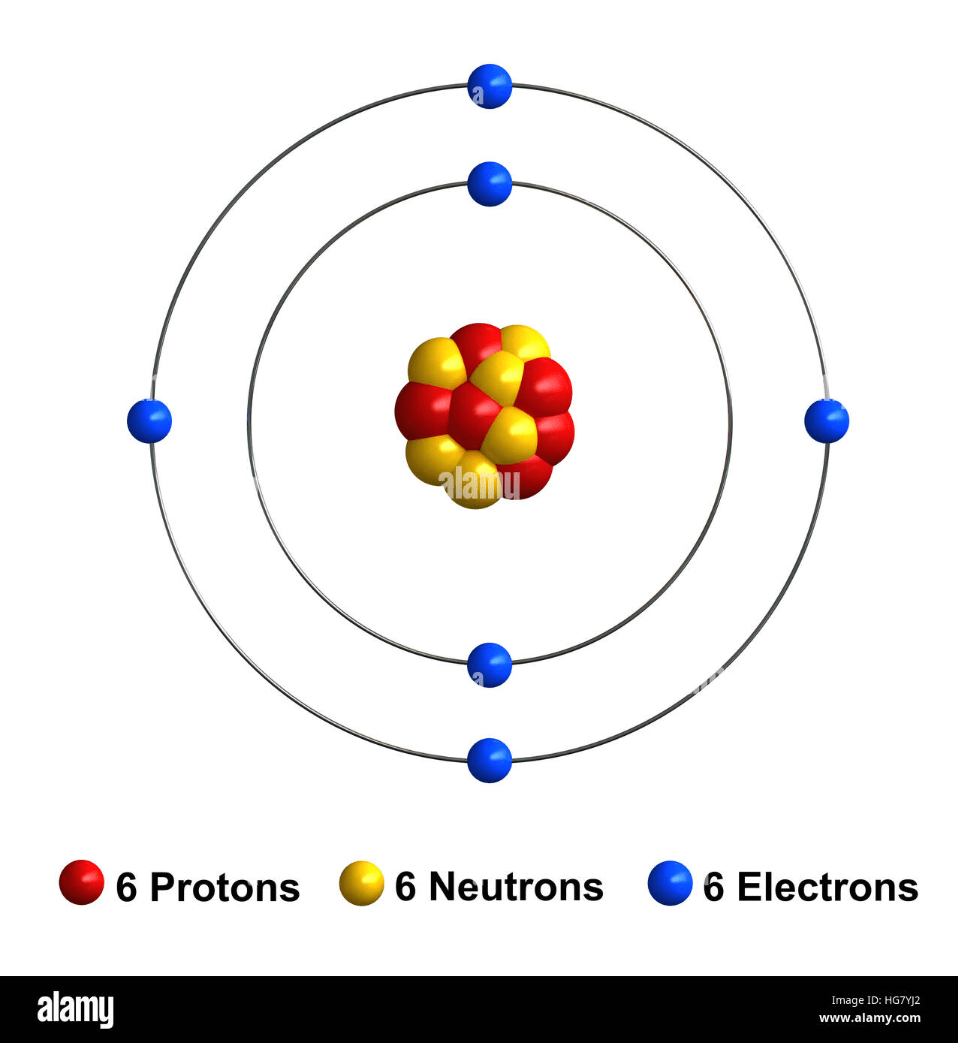
4 electrons
How many protons, electrons, and neutrons in a Zinc atom?

30 protons
30 electrons
35 neutrons
What sport did Mr. Schmid flunk out of in 6th grade?
a. Gymnastics
b. Baseball
c. Soccer
d. Tennis
Baseball
What are two examples of things NOT made of matter?
Answers may vary.
Possible answers: Energy, Thoughts, Love, Light, Sound, Friendship, etc
Why of the atoms below will be more reactive and WHY?

The atom on the right
The right atom only has one valence electron, making its outer shell almost empty. Atoms with nearly empty shells are very reactive.
The left atom has an outer shell with 4 electrons, making it halfway between full (8) and empty (0). Therefore, it is not very reactive.
Electrons fly around the nucleus so quickly that if an electron were to travel around the entire Earth, it would take only 18 seconds!
Why do an atom's electrons stick around the nucleus instead of just flying away?
Opposite charges attract!
Electrons have a negative charge and so they are attracted to the positive charge of the proton(s) in the nucleus.
What element is this?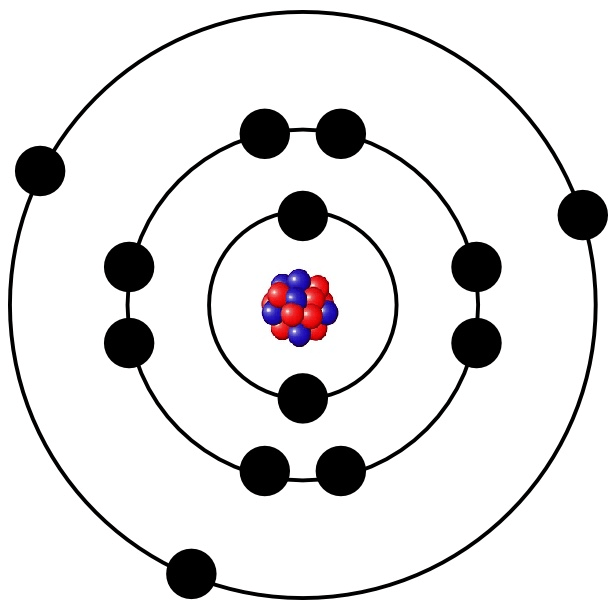
Aluminum
What two bones has Mr. Schmid broken?
a. Nose and Leg
b. Tailbone and Arm
c. Rib and Foot
d. Finger and Skull
Tailbone and Arm
The word "atom" comes from the Ancient Greek ἄτομος (átomos), which means..... what?
Uncuttable
Indivisible
These are atom from three Noble Gases - Helium, Neon, and Argon. Based on what you know about atomic structure, why are they called "noble" gases?
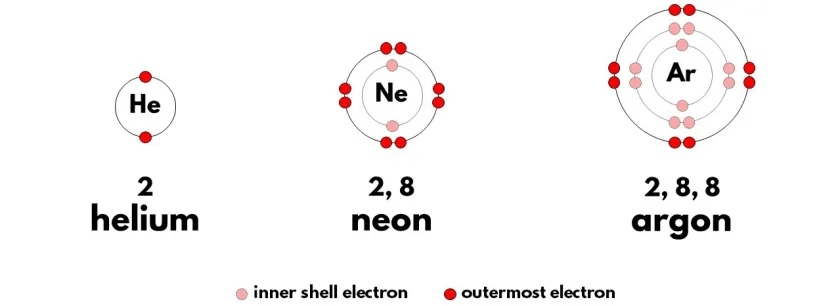
These elements are "Noble" because with full outer shells, they are not reactive and are stable, reliable, safe.
Aka Noble.
What are the forces cause atoms to bond together?
Atoms bond together because opposite charges attract.
The positive proton in one atom is attracted to the negative electrons in another atom.
What element is this?
Argon
What is the only animal that Mr. Schmid fears?
Leeches
