These are known as the fundamental "building blocks" of all matter.
What are atoms
In the chemical formula CO2, this is the total number of different elements present.
What is two? (Carbon and Oxygen)
These contain only one specific type of atom.
What are elements
Scientists use these to describe the atomic composition of simple molecules and compound molecules.
What are models?
Besides having mass, atoms also have this physical property, meaning they "take up space."
What is volume
This is the total number of individual atoms found in a single molecule of CO2
What is three
These are formed when different types of atoms are chemically combined.
What are compounds?
In a model of H2O what element does the central atom represent
What is Oxygen
To give you an idea of how small they are, atoms are significantly smaller than these human body cells.
what are red blood cells
The formula O3 represents this type of molecule because it only contains one type of atom.
What is an elemental molecule?
This molecule would be classified as a(n) 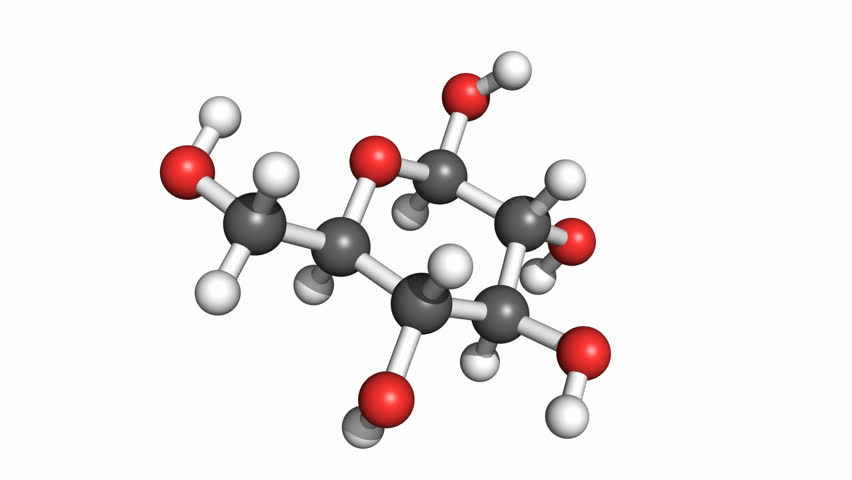
What type of molecule is represented by the picture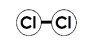
What is an elemental molecule?
When two or more atoms bond together, they form these.
What are molecules?
Common substances like H2O and CO2 are categorized as this because they contain different types of atoms.
What are compounds?
This substance, abbreviated as He, is classified as an atom rather than a molecule.
What is Helium?
When looking at a model, you can tell it is a compound if it has atoms of different sizes or these.
What are different colors (or types)
True or False: Atoms are the smallest unit of an element that maintains the properties of that element.
What is an true?
This is the chemical formula for Glucose, a complex molecule that we have spoken about. What are the three elements that make it up?
C6H12O6
What is Carbon, Hydrogen, Oxygen
A molecule like O3 is classified as a molecule because of this specific reason.
What is it consists of more than one atom bonded together?
This is the main difference between how elements and compounds are modeled.
Elements show one type of atom while compounds show multiple types