This property means a material is good and allowing heat to transfer through it quickly
Conductor
How many atoms are there in glucose?
C6H12O6 ?
24 atoms!
6 Carbon
12 Hydrogen
6 Oxygen
The name for the 1 or 2 letter abbreviations on the periodic table that stand for each element.
Chemical Symbol
This Scientific Law explains how energy is not lost when changing one form to another
Law of Conservation of Energy
The type of circuit in this image is
A. Parallel
B. Series
C. Neither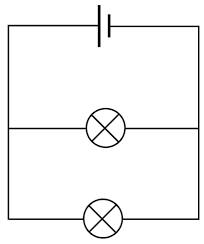
Parallel! There is more than one path for electricity to flow
The Law of Conservation of Mass tells us that
the mass will stay constant before & after any reaction, no mass is created or destroyed
Which of these equations is balanced?
1. H2O -> H2 + O2
2. 1F2 + 1Na -> 1NaF
3. 2Mg + Si -> Mg2Si
Equation 3 is balanced
The vertical columns are called
Groups / Families
This type of nuclear reaction takes place in the sun and other stars
Fusion
A disadvantage of an electromagnet over a permanent magnet includes _______________________
Strong ones are expensive
A constant power supply is needed or it goes off
What is the name of the scientific theory that explains how matter gains more energy and can change states of matter if they gain enough energy
Kinetic Molecular Theory
name the two groups from the periodic table that frequently make ionic bonds together
Metals & nonmetals
The horizontal rows are called
Periods
When a cold blooded lizard basks in the sun to warm up, it receives thermal energy via ________
Radiation
 If you charge an object via conduction, what is it that you must do?
If you charge an object via conduction, what is it that you must do?
Touch the materials together
This word describes a reaction that absorbs heat from the surroundings, usually making it feel cold
Endothermic (Endo is inside, so heat goes into it)
covalent bonds typically form between ___________ & _____________
nonmetals & nonmetals
Calcium (Ca) is in Group 2 & Period 4. It shares more in common with which elements:
1. The elements in Group 2
or
2. The elements in Period 4
Group 2!
The maximum potential energy someone on a playground swing experiences is where?
at the top of the swing arc!
Height & gravity & mass are the things that matter for gravitational potential energy.
Mass and gravity are unchanged here = only height is changing
Which of these is a resistor?
-Battery
-lightbulb
-wires
-iron core
Lightbulb! Any device that absorbs power from the circuit is a resistor, with some of them having a purpose of adding electrical resistance to slow current in a circuit, like the one in the image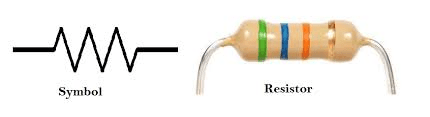
which is more dense?
water
OR
water after you add salt
why?
.
water after you add salt, because there is more matter in the same sized amount of water, thanks to dissolved salt
This vocab word is a mixture that looks the same throughout, like milk or orange juice, and has dissolved solids. Using evaporation is one of the ways to separate the matter.
Homogeneous mixture
What does the zigzag line on the periodic table mean?
It separates metals & Nonmetals (Metalloids are found above or below this line, and we will discuss this later)
The order of energy types in this scenario: Sunlight hits a leaf, conducts photosynthesis, a person eats the leaf in a salad, powers their brain
Radiant - Chemical - Electrical
Magnets always have this many poles, no matter the shape? What are they called?
2 (North & South)