This is how organisms transfer their genetic information from one generation to the next
Reproduction
Polymers are large chains made from repeating these simple molecules
monomers
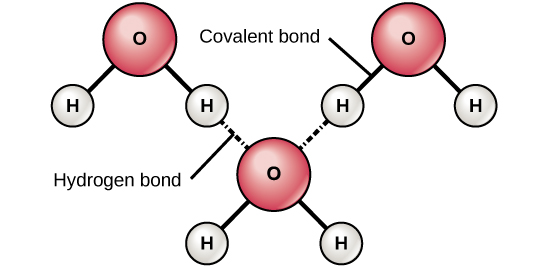
Molecules of water are held together by a strong force. The property of water that causes it to have a high boiling point compared to other small molecules is due to the presence of this kind of bond between molecules
hydrogen bonding
What is X?

enzyme
This is an educated guess about a scientific phenomenon that is based on observations and previous knowledge
hypothesis
Pulling your hand away from something hot, or a plant growing towards sunlight is an example of this characteristic of life
response to stimuli
This macromolecule consists of sugars such as glucose and provides short term energy.
carbohydrates
Which properties are caused by hydrogen bonds between the slightly negative side of one water molecule and the slightly positive side of another water molecule, holding water molecules together
cohesion & surface tension

Most enzymes acts as this, speeding up a reactions without becoming part of the products
catalyst
This is a well-substantiated explanation of some aspect of the natural world that is acquired through the scientific method and repeatedly tested and confirmed, preferably using a written, pre-defined, protocol of observations and experiments.
scientific theory
This molecule is the most important for cells getting stuff done. It is made through cellular respiration and metabolism.
ATP - adenosine triphosphate (energy)

This macromolecule is responsible for building hair, skin, nail health, and helps your immune system. The enzymes speed up reactions.
Proteins
Water can absorb a lot of heat before its temperature changes. This property helps regulate temperature on Earth
Changes in temperature or pH can cause an enzyme to change shape and function. What do we call this?
denature
In an experiment, this is the "normal" group that provides a standard for comparison
control group
Organisms will adapt or change over time with the environment due to natural selection as part of this process
Evolution
These are important in the cell membrane because they have both a polar (hydrophilic) and a nonpolar ends (hydrophobic). They store energy for a long period of time.
Lipids
If you eat too much sugar and feel sick, you should drink water because it is a...
universal solvent - because of its polarity
Which row in the chart below contains correct information concerning fast synthesis of molecules?

3
An experiment is carried out to determine how different pH values of soil will affect the growth of tomato plants. In this experiment, the dependent variable could be the...
height of the tomato plants
A lizard spends the morning lying in the sunlight until its body temperature rises. Later in the day, it rests in a shady area until its body temperature cools. This type of behavior is important to maintain
homeostasis
All biological macromolecules are made up of carbon, oxygen, and hydrogen. Proteins also contain nitrogen and this class of macromolecules contain phosphorus as well. It is in all cells.
nucleic acids - DNA and RNA
Water’s ability to form hydrogen bonds contributes to this characteristic that allows organisms to stay cool during evaporation, such as through sweating or transpiration in plants.
evaporative cooling

The graph represents the effect of pH on 3 different enzymes at normal body temperature.

The graph illustrates enzymes 1, 2, and 3. Which one represents an acid?
Enzyme 1 = pH of 3
A student wanted to investigate the effect of light on the rate of ripening of tomatoes. She set up four pots of the same size with identical amounts of soil, water, and type of tomato plants. Each plant was exposed to a different intensity of light as shown in the table below.

What is the IV?
light intensity