Which element has 56 protons?
Barium
How many electrons does zirconium have?
40 electrons
What data does a mass spectrometer provide
mass and abundance for each isotopes
what determines the position of an element in the periodic table ie period and group
number of protons (atomic number)
and electron arrangement (energy levels and valence electrons)
What causes an absorption spectra?
Electrons absorbing specific energy to move up energy levels
Which element has 16 as its mass number?
oxygen
How many electrons does the ion of sulfur have?
18 electrons
S2- = 16 + 2
What affects the amount of deflection for an atom?
its mass and charge
a smaller mass deflects more and
a greater charge deflects more
What is the order from largest to smallest radi?
Potassium Bromine Francium
Francium, Potassium, Bromine
Does a flame test provide qualitative or quantitative data?
qualitative data
How many p, n, e does the atom P-31 contain?
p = 15, n= 16, e = 15
Give the electron configuration for nickel?
2, 8, 16, 2 or
1s2 2s2 2p6 3s2 3p6 4s2 3d8
Determine the relative mass for nitrogen with 2 isotopes; nitrogen-14 (N-14) at about 99.62% abundance and nitrogen-15 (N-15) at approximately 0.38% abundance
Ar(N) = (14x99.62) + (15x0.38) / 100
= 14.0038
What element has 3 valence electrons and 4 energy levels?
Gallium
Why does Lithium produce a different emission spectra to sodium?
Each element has a unique spectra based on the specific energies required for electrons to fit into its energy levels
What is the identity, charge and mass number of the atom with p = 53 n=74 e =54
Iodine ion, mass 127, with a -1 charge
Draw the Lewis structure for selenium
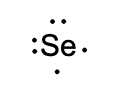
What are the steps in order that occur in mass spectrometry
vaporise (become a gas), ionise (become charged), accelerate (increase KE), deflect (by electromagnet), detect (mass and abundance data)
Identify the trend for radius, ionisation energy and electronegativity going down a group
radius increases
ionisation energy and electronegativity decrease
What does AAS stand for?
Atomic Absorption Spectroscopy
Determine the p, n, e for Al3+ ion with a mass of 27
p = 13, n= 14, e = 10
Give the electron configuration for strontium
2, 8, 18, 8, 2 or
1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p6 5s2
Copper has 2 isotopes Cu 63 and Cu 65. Which has the greater abundance?
Cu 63 (as the relative mass = 63.55)
Why does Fluorine have a large electronegativity?
it has a stronger attractive force to attract other atoms electrons due to its large core charge and small radius (force acts over a shorter distance)
What is used to create a calibration curve?
the absorbance readings from a set of standard solutions for the element being measured