AZX
Label the components above
A = mass #
Z = atomic #
X = symbol of elementIs blood a heterogeneous or homogeneous mixture? Why?
Heterogenous; it is not uniformally distributed and can be separated non-chemically (by centrifuge)
What is the mass of 50.0 mL of octane, with a density = 0.702 g/cm3?
35.2 g
What are the charges and placement of subatomic particles?
Neutrons and protons in the nucleus, electrons in a cloud around the nucleus; protons have +1 charge, electrons -1, neutrons neutral
BONUS: Which have notable mass?
If wavelength decreases, frequency ___
increases
(T/F) isotopes are atoms with the same atomic # but different mass #
(T/F) isotopes of the same element are equally abundant in nature
1) True
2) False
What is the difference between an element, a mixture, and a compound?
element - identical atoms
mixture - nonbonded components
compound - chemically bound components
Convert 20.5 cm3 to m3.
2.05 x 10-5 m3
Given the ion with a 3+ charge, 28 electrons, and a mass number of 71, write out the isotope formula
7131Ga3+
Which parts of the quantum wave relate to energy?
(Frequency, Amplitude, Wavelength)
Frequency and wavelength
The group # on the periodic table corresponds to _____
Number of valence electrons
Which of the following scenes depict pure substances?
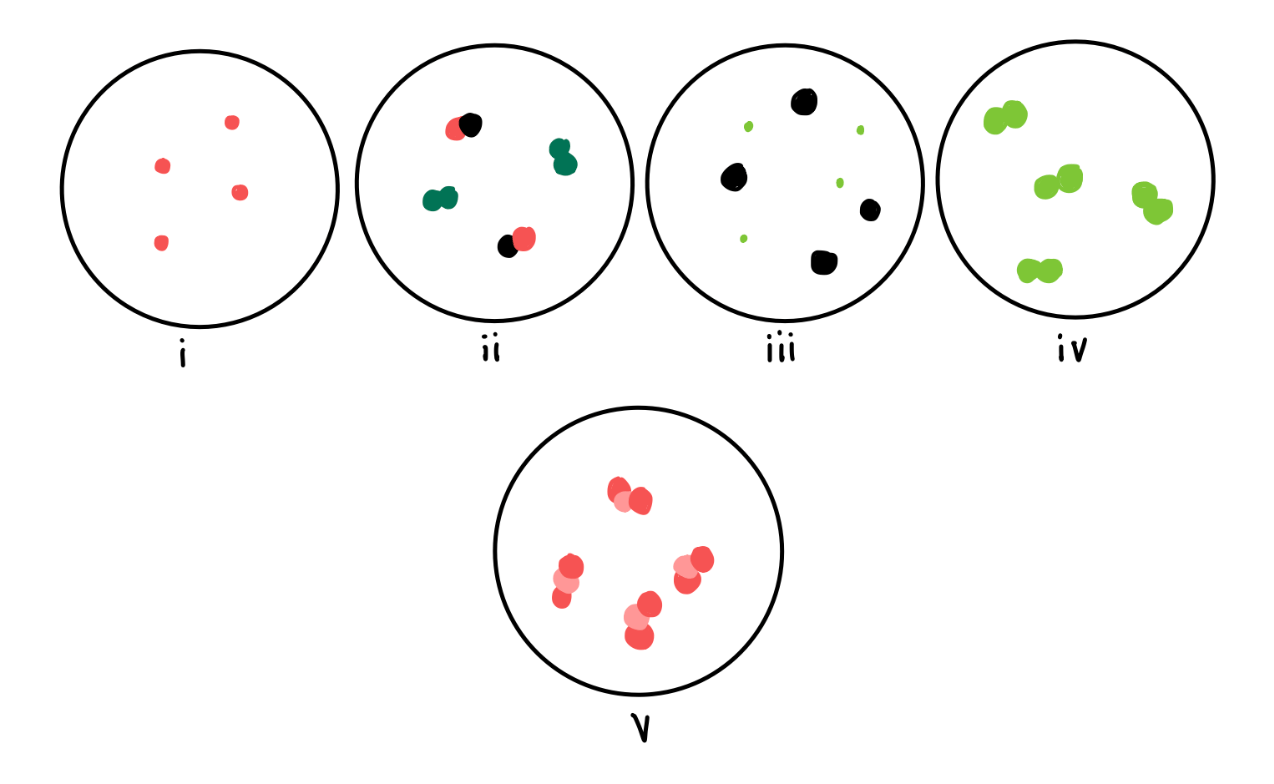
i, iv & v
The density of a certain substance is 6.3 g/cm3. Aliens have landed on your planet and demand that properties be expressed in their units of density, glorps/eeks3. 1 glorp = 0.324 g, and 1 cm = 2.7 eeks. What is the density in alien units?
9.9 x 10-1 glorps/eeks3
Which isotope has the greatest number of neutrons?
a) chlorine-36
b) carbon-14
c) silicon-29
a) chlorine-36
What element is 1s22s22p3? How many valence electrons does it have?
Copper has a density of 8.96 g/cm³ near room temperature. A piece of copper weighs 5.6 mg, has a length of 6.8 cm, and a width of 3.4 cm. What is its height in millimeters (mm)?
0.00027 mm
Classify the six italicized properties in the following paragraph as chemical or physical:
Fluorine is a pale yellow gas that reacts with most substances. The free element melts at −220 °C and boils at −188 °C. Finely divided metals burn in fluorine with a bright flame. Nineteen grams of fluorine will react with 1.0 gram of hydrogen.
Pale yellow: physical
Gas: physical
Reacts with most substances: chemical
Melts at -220 oC: physical
Boils at -188 °C: physical
Metals burn in fluorine: chemical
Nineteen grams of fluorine will react with 1.0 gram of hydrogen: chemicals
If there are 688.35 mg of a substance in a volume of .0560 L, what is the substance's density in g/mL?
0.0123 g/mL
Which of these atoms has the greatest number of neutrons in its nucleus?
a) 5727Co
b) 2814Si
c) 5625Mn
d) 5526Fe
c) 5625Mn
1s22s22p6 with an ionization of +2 charge would correspond to ____
Magnesium
The element antimony is made up of two isotopes, 121Sb with a mass of 120.9038 amu and 123Sb with a mass of 122.9042 amu. Given that the average atomic mass is 121.760 amu, what are the percent abundances of the two isotopes?
121Sb is 0.5720, and percent abundance is 57.20 %.
Fractional abundance of 123Sb is therefore 0.4280, and percent abundance is 42.80 %
If a shiny iron nail with an initial mass of 23.2 g is weighed after rusting, would you expect the mass to have increased, decreased, or remained the same? Explain
BONUS: Is this a physical or chemical change?
Increased as it would have combined with oxygen in the air, thus increasing the amount of matter and therefore the mass.
BONUS: Chemical
As an instructor is preparing for an experiment, he requires 225 g phosphoric acid. The only container readily available is a 150-mL Erlenmeyer flask. Is it large enough to contain the acid, the density of which is 1.83 g/mL?
Yes, because the acid's volume will be 122.95 mL
What are the diagrams below representing?
BONUS: What is an orbital?

Left (s orbital), right (p orbital).
BONUS: The probability cloud of finding an electron
1s22s22p1