The first person to come up with the term for an atom was _______________.
Democritus
This model of the atom is just a sphere.
Dalton's model- Billiard Ball Model
What is the FULL electron configuration for chlorine
1s2 2s2 2p6 3s2 3p5
This scientist applied the principles of quantum physics to the spectrum of hydrogen gas to develop a popular model of the atom.
An ion is at atom that has gained or lost _______________.
Electron(s)
The scientist to introduce the idea that atoms had a nucleus surrounded by electrons spinning in empty space was ________________.
Rutherford
This model of the atom has a dense positive nucleus with electrons floating around the outside
Rutherford model- Nuclear model
What is the CORE notation for tin (Sn)
[Kr]5s2 4d10 5p2
This scientist proposed that energy is not just a wave, but can also behave like particles in the form of energy packets called quanta.
Max Planck
Give the number of protons, neutrons, and electrons for Tungsten - mass number is 184
74 protons, 110 neutrons, 74 electrons
The modern day atom describes the electrons existing in an __________ ________.
electron cloud
This model of the atom is a positively charged sphere with negatively charged electrons dispersed throughout.
Thomson model-Plum Pudding Model
Write the core notation for Chromium (Cr)
[Ar] 4s1 3d5
This scientist developed the wave equation that allows us to map atomic orbitals. This equation allows us to predict the region of space where the probability of finding an electron is the highest.
Erwin Schrödinger
What is the difference between the white light spectrum and the hydrogen gas spectrum?
The white light spectrum is the continuous spectra/bright line emission spectra/all the colours of the rainbow are visible
Hydrogen gas spectrum is a discontinuous spectrum- only see violet, green, and red- as electrons "jump" and "fall" they release a certain wavelength depending on the shell since the energy between shells is fixed
Name the scientists who discovered the proton, neutron, and electron (i.e. _____ discovered the _______)
1. electron - JJ Thomson 1897
2. proton - Rutherford 1912
3. neutron - Chadwick 1932
This model of the atom put electrons in circular orbit energy levels around the nucleus.
Bohr model- Planetary Model
Write the core notation for Zr2+ (think ptsd)
[Kr] 4d2
This scientist was the first to propose that particles of matter (like electrons) exhibit wave-like properties
Louis De Broglie
Find the average atomic mass for copper in amu (round to the third decimal place)
Isotope Percent Abundance Mass (amu)
Copper-63 69.17% 62.930
Copper-65 30.83% 64.928
63.546 amu
Name the statement for the atomic theory that was the same for ideas from Democritus and John Dalton.
All matter is made up of atoms that cannot be divided, created, or destroyed.
Which scientist created the Mechanistic Model?
Democritus
Write the number of valence electrons for indium (In) (hint* write electron configuration first)
3 valence electrons
[Kr] 5s2 4d10 5p1
This scientist showed that it is impossible to know both the exact position and it’s momentum at the same time.
Werner Heisenberg
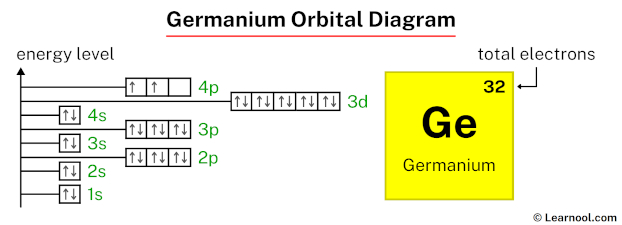
Draw the orbital diagram for germanium (Ge)