What is the speed of light?
3.0 x 108 m/s
What is the unabbreviated ground state electron configuration for Zirconium (Zr)
1s² 2s² 2p⁶ 3s² 3p⁶ 4s² 3d¹⁰ 4p⁶ 5s² 4d²
What element has the largest atomic radius?
Francium
One meter is equal to how many nanometers?
1 x 109 nanometers
What university did Mr. Dame go to?
Heidelberg University
What is the wavelength of a wave with a frequency of 5.46 x 1016 Hz?
5.48 x 10-9 m
What is the abbreviated ground state electron configuration for Silicon
[Ne]3s23p2
Which element has the highest ionization energy?
Helium
Which atom is expected to have a half-filled 5s subshell?
Rubidium
What salt made a red flame during our flame test lab?
Strontium Chloride
The laser on a Blu-ray player has a wavelength of 405 nm. What is its frequency?
7.38 x 1014 Hz
Which atom would be expected to have a half-filled 6p subshell?
Bismuth
What type of relationship does Atomic radius and ionization energy trends have?
Based on their positions in the periodic table, list the following atoms in order of increasing radius: Mg, Rb, Cs, Ca.
Mg < Ca < Rb < Cs
How do solar panels work?
The light from the sun has enough energy to break electrons of of the thin sheet of silicon that make up the panel and that free electron is transferred into electricity.
How much energy does a photon of light have if it has a frequency of 1.85 x 105 Hz?
1.23 x 10-28 J
1s22s22p63s23p64s23d7 is the grounds state electron configuration for which element?
Co - Cobalt
Based on their positions in the periodic table, predict which has the largest first ionization energy: Mg, Ba, B, O, Te.
O
Determine the atom being represented in the bohr model below.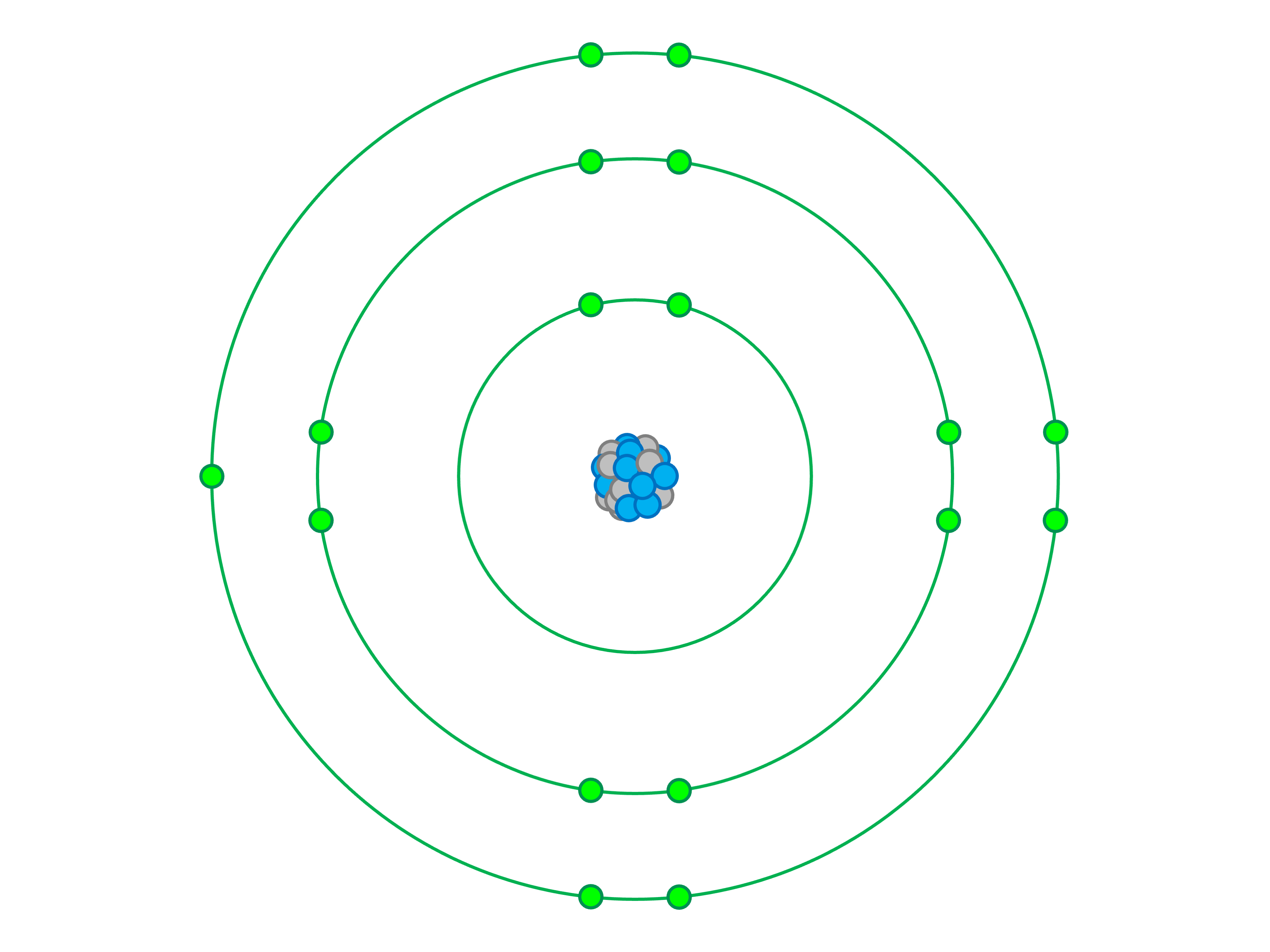
Chlorine
What does Mr. Dame's wife do for a living?
Mobile dog groomer
Heated lithium atoms emit photons of light with an energy of 2.961 × 10−19 J. Calculate the frequency and wavelength of one of these photons.
Frequency= 4.47 x 1014 Hz
Wavelength= 6.68 x 10-7 m
Write the unabbreviated electron configuration of Co3+.
1s22s22p63s23p64s23d4
Based on their positions in the periodic table, list the following ions in order of increasing radius: K+, Ca2+, Al3+, Si4+.
Si4+ < Al3+ < Ca2+ < K+
Light with a wavelength of 525.3 nm looks green. What is the energy, in joules, per photon of this green light?
3.77 x 10-19 J
How many times does Mr. Dame say his "Alright" intro in a 5 day week?
30 times! 6 times a day, five days a week! Making sure you all are having great days!