What did Niels Bohr do?
He developed the first simplified model of an atom.
Chemical symbols and numbers abbreviating the name of a compound are what?
A chemical formula
List two characteristics and one example of an acid.
Lemons, vinegar, tomato juice
Sour taste
Can burn
corrosive
The study of matter
Chemistry
If an element has 72 protons, how many electrons does it have? Why?
72, because a normal atom has the same number of electrons and protons.
What is the range of the pH scale?
1-14 (or 0-14)
What do you get when you add together an atom’s protons and neutrons?
The atom’s atomic mass
Describe a covalent bond.
When atoms share their electrons.
List two characteristics and one example of a base.
Slippery to touch
Bitter to taste
baking soda, soap, bleach
A substance that is only made up of one kind of atom
Element
What are two things you can tell about an element by where it is on the periodic table?
The periods tell you how many shells of electrons the element has and the groups tell you how many electrons are in the element’s outer shell.
This is the same as the amount of protons an atom has.
Atomic number
Describe an ionic bond.
When positively and negatively charged ions attract.
True or False:
A substance that changes color when it is exposed to an acid is called a ion.
False, indicator.
An atom that has gained or lost electrons
Ion
How many Hydrogen molecules are in water?
TWO
A substance that is made up of only one type of atom.
element
What kind of reaction breaks compounds down into simpler compounds?
Decomposition Reaction
How can you determine the concentration of acids or bases in a solution?
By using the ph scale.
An ionic compound produced from a reaction between an acid and a base
Salt
Explain why all compounds are molecules but not all molecules are compounds.
There are different types of molecules. Some molecules are produced when atoms of the same element combine. Others are produced when atoms of two or more different elements combine through a chemical reaction. Compounds are a kind of molecule produced as a result of a chemical change.
Draw and label an atom with the following parts:
Electron, proton, nucleus & neutron

How many molecules of Oxygen are in the following compound?
(Bonus points if you know the common name)
C12H22011
11
SUGAR-Bonus $200
What is something that you could use from your house (think of supplies from the kitchen or bathroom) to relieve the pain of a mosquito bite? Why?
Any answer that has to do with a base neutralizing an acid is correct!
Ex: baking soda, toothpaste, milk of magnesia, etc.
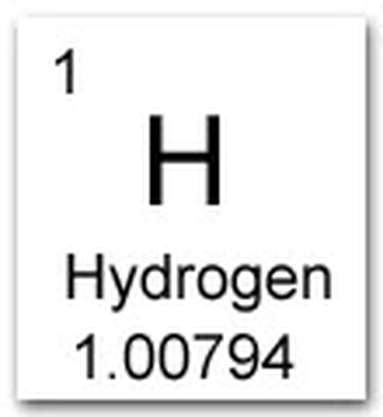
Draw and label the element square for Hydrogen.
Hints:
H, 1, 1.00794
What are noble gases considered as?
Hint: Full outer shells
Stable