What kind of reaction is this?
CaCO3 → CaO + CO2
Decomposition Reaction
Is this reaction exothermic or endothermic?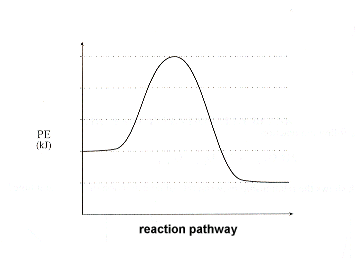
Exothermic
H2 + O2 --> H2O
2H2 + O2 --> 2H2O
What is the molar mass of Lanthanum (La)?
138.91 g
Aluminum reacts with ferrous oxide to form iron and aluminum oxide.
Which substances are the reactants?
Aluminum and ferrous oxide
What kind of reaction is this?
8 Fe + S8 → 8 FeS
Synthesis Reaction
Is this reaction exothermic or endothermic?

Endothermic
P4 + O2 --> P2O5
P4 + 5O2 --> 2P2O5
How many particles of a substance are there in a mole?
6.022 x 1023
In this redox reaction, hydrogen is oxidized. What must be true about fluorine?
H2 + F2 → 2HF
Fluorine is reduced
Which kind of reaction involves a fuel reacting with oxygen?
Combustion reaction
If you mix baking soda and vinegar together and touch the side of the container that the reaction takes place in, you will notice that it becomes colder. What word is used to describe this?
Endothermic
NO + O2 --> NO2
2NO + O2 --> 2NO2
Create a conversion factor for Rubidium (Rb) (moles and grams)
1 mol Rb/85.47 g Rb
OR
85.47 g Rb / 1 mol Rb
A system is at equilibrium with the following reverse reaction happening:
NH4Cl ⇌ NH3 + HCl
If a bunch of NH4Cl is added to the mixture, what will happen according to Le Chatelier's principle?
A bunch of NH4Cl will react in order to return to equilibrium
What kind of reaction is this?
2AgNO3 + Cu --> Cu(NO3)2 + 2Ag
Single displacement reaction
What does the law of conservation of energy state?
Energy cannot be created or destroyed; it can only change forms
H2O2 --> H2O +O2
2H2O2 --> 2H2O + O2
In the following chemical equation, how many moles of SO2 are produced?
S8 + 8O2 --> 8SO2
8 moles
Why does heating up a substance cause it to react more readily with other substances?
3 criteria for reaction are:
- particles collide
- With proper alignment
- With enough energy
Heating a substance gives it more energy, increasing chances of reactions happening.
What kind of reaction is this?
2KI + Pb(NO3)2 --> 2KNO3 + PbI2
Double Displacement Reaction
In this situation, what role does the focused sunlight play?
It provides activation energy for combustion to take place.
SiCl4 + H2O --> SiO2 +HCl
SiCl4 + 2H2O --> SiO2 + 4HCl
In the following chemical equation, how many moles of hydrogen (H2) react with nitrogen to form 2 moles of nitrogen hydroxide?
3H2 + N2 --> 2NH3
3 moles of hydrogen
What is wrong with this chemical equation?
2F3 + 3Li2SO3 --> B2(SO3)3 + LiF
Boron (B) is missing from the reactants side of the equation - this breaks the law of conservation of matter