What are IMFs?
An IMF are attractive forces that exist between ALL particles.
What is it called when a solid changes to a gas?
How about a gas to a solid?
Sublimation
Deposition
Add in addtional lone pairs and hydrogen atoms to molecule below:

*instructor will draw correct molecule on board*
Relate y=mx+b to (ln P= -ΔHvap/-RT = ln A)
ln p= y axis
x= 1/T
b= ln A
m= -ΔHvap/-RT
Why does boiling point increaes as molar mass increases?
Ex) Fluroine has a molar mass of about 38g/mol and a boiling point of -188°C, wheras Chlorine has a molar mass of 35g/mol and a boiling point of -34°C
Boiling point increases as molar mass increaes because a greater molar mass indcates the e- are further from the nucleus therefore more polarizable*
*polarizable is a fancier way of saying they make more temporary dipoles
List the three types of IMFs in order of weakest to strongest.
dispersion forces<dipole-dipole<hydrogen bonds
What is the difference between endothermic reactions and exothermic?
Exothermic reactions release energy into surrounding (negative value). The system goes from high energy to low energy.
Draw the skeletal structure for the following molecule:
CH3CH2CH2OH
*instructor will draw on board
How are IMFs related to viscosity?
If a substance is more viscous, it will have stronger IMF's
List all IMFS and why for this molecule:
Dispersion: all molecules have dispersion
Dipole-Dipole: from polarity
List all IMFs that this molecule has and why:

dispersion forces and dipole-dipole.
Dispersion because all molecules will have dispersion attraction.
Dipole-dipole due to the polarity of the oxygen molecule
What is vapor pressure? Why does it occur?
It occurs due to liquid molecules bumping into one another and becoming a gas
How many carbons and implied hydrogens are on the molecule below:
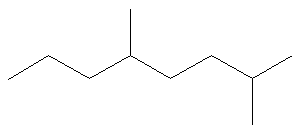
Carbons: 10
Hydrogens: 22
Explain the two forces that occur during capillary action:
cohesive force: molecule attraction wihtin the liquid
adhesive force: molecule attraction between liquid and tube
Answer these:
Solids tend to be present at ___ temp and ___ pressure.
Liquids tend to be present at ___ temp and ___ pressure.
Gases tend to be present at ___ temp and ___ pressure.
Solids: low and high
Liquids: high and high
gases: high and low
What is the difference between hydrogen bond acceptors and donors?
A hydrogen bond acceptor will posses a N or O atom with lone pairs.
Hydrogen bond donors will contain a N-H or O-H bond.
Define enthalpy of vaporization:
Draw the graph and label axes
energy needed to take 1 mol of a liquid and make it a gas (at constant P and T)
*instructor will draw graph
Identify all functional groups on the following molecule:
alcohol and carboxcylic acid
What is surface tension?
Liquids reducing their surface area, this generates stronger IMF's.
List the functional group shown in this molecule:
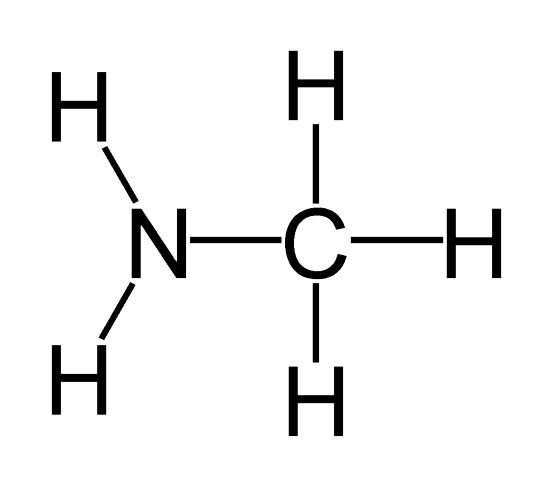
amine
Diethyl ether and n-butanol have the exact same molar mass (74.12 g). Explain why diethyl ether has a significanly lower boiling point (35°C) than n-butanol (117°)?
The difference in boiling point comes from n-butanol containing dispersion, dipole-dipole, and hydrogen bonds. Whereas diethyl ether has dispersion and dipole-dipole.
How is vapor pressure related to the IMF strength between molecules?
If a molecule has a low vapor pressure, then it's intermolecular forces are strong.
Functional groups are important because they alter the physical and chemical properties of a molecule. This will affect reactions
Draw a phase change diagram. Label the point where solids, liquids, and gases all exist. Additionally, label the line where vapor pressure exists.
*insturctor will draw answer
How many carbons and hydrogens are in this molecule:
Carbons: 5
Hydrogens: 9