Group 7A element
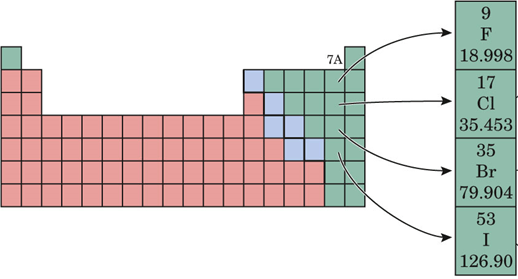
What is a halogen?
Atomic mass of a proton
What is 1 amu?
Assumes the shape of it's container and is highly compressible
What is a gas?
Mass divided by volume
What is density?
Element C
What is carbon?
The columns on the periodic table
What are families?
Number of electrons in 2 p orbitals
What is 6?
Well defined shape and essentially in compressible
What are solids?
Amount of heat needed raise 1 gram of water, 1 degree Celcius
Element O
What is oxygen?
Group 1A elements
What are alkali metals
Exists in the nucleus
What are protons and neutrons?
Iron and sulfur combined and and each component is visible
Energy in chemical bonds
What is potential energy
Atom with the smallest radius
What is F?
Elements on the left side of the periodic table
What are metals?
Occurs outside of the nucleus
What are electrons?
Weighted average of isotopic masses
What is atomic weight?
1000 mL
What is 1 L?
126C and 136C are an example of
What are isotopes?
Location of nonmetals on the periodic table
What is the right side?
Number identifying the element
What is atomic number?
Matter can neither be created nor destroyed.
What is Law of Conservation of Mass?
Typically solid and conducts electricity
What is a metal?
How Hydrogen, Oxygen, Nitrogen, Fluorine, Chlorine, Bromine and Iodine exist in nature
What are the diatomic elements?