The classification of the following amine
What is primary
Classification of the following amide

What is primary
The common name for the following compound

What is propanoic acid
The product formed when water gains an extra hydrogen
CH3COOH + H2O -----> ?
What is the hydronium ion
Behavior of amines when dissolved in water
What are bases
Another acceptable name for benzenamine.

What is aniline
Classification of the following Amide

What is secondary
The IUPAC name of the following compound?
What is 3-chlorobutanoic acid
The product of an acid base reaction of Acetic Acid and Potassium Hydroxide
CH3COOH + KOH
What is potassium acetate
Product formed from CH3NH2 in water
What is methylammonium ion
The systematic name of the following compound
What is N,N-dimethylmethanamine
IUPAC name for the following compound
What is N-propylbutanamide
Systematic name of the following compound
What is ethyl pentanoate
propanoic acid reacts with potassium hydroxide
What is potassium propanoate
Product of RNH2 (base) + HCl (acid)
What is an alkylammonium salt
The IUPAC name of the following compound

What is N-methylpentan-2-amine
IUPAC name of the following compound
What is hexanamide
IUPAC name of the following carboxylic acid derivative

What is acetic anhydride
The salt formed by the reaction of butanoic acid and sodium hydroxide.
What is sodium butanoate
Product of an amine reacting with water
what is an alkylammonium ion
The IUPAC name of the following compound

What is N-butylbenzamide
IUPAC name of the following compound
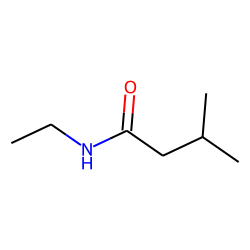
What is N-ethyl-3-methylbutanamide
IUPAC name of the following carboxylic acid derivative

What is 3-methylpentanoyl chloride
The product of an acid-base reaction of carboxylic acid in water
CH3COOH + H2O -----> ?
What is the carboxylate anion
The chemical behavior of amines
what are weak bases
