(2 min) The work function of copper is 7.53 x 10-19 J. Calculate the energy of an electron ejected at half of the maximum velocity by irradiation with a wavelength of 77.2 nm. The mass of an electron is 9.11 x 10-31 kg.
4.56 x 10-19 J
(3 min) If 0.450 M sodium azide (NaN3) is added to water at 25ºC. If the Kb of hydrazoic acid (HN3) is 1.9 x 10-5, please determine the resulting pH.
pH = 9.19
(2 min) If the enthalpy of combustion of some hydrocarbon “X” is -2877 kJ/mol and the entropy of combustion is -1.488 kJ/Kmol, please calculate the range of temperatures at which this reaction is spontaneous.
T <= 1933 K
(3 min) Balance the following redox reaction in basic conditions:
PbO (s) + ClO3- (aq) → Cl2 (g) + PbO2 (s)
5 PbO + H2O + 2 ClO3- → Cl2 + 2 OH- + 5 PbO2
(2 min) Radon-222 has a half-life of 4 days. How long does it take for 83% of radon-222 from a sample to decay?
8.83x105 s = 10.23 days
(2 mins) Derive the Henderson-Hasselbalch equation from the Ka expression of a generic weak acid “HA”. When should you use this equation?
Ka = ([H^+][A^-])/([HA])
pKa = -log(Ka) = -log(([H^+][A^-])/([HA])) = -log(H^+) - log([A^-]/([HA]))
pKa = pH - log([A^-]/([HA])) = pH + log([HA]/([A^-]))
pH = pKa - log([HA]/([A^-]))
(3 mins) Solve for the radial and angular nodes for a hydrogen 3px orbital.
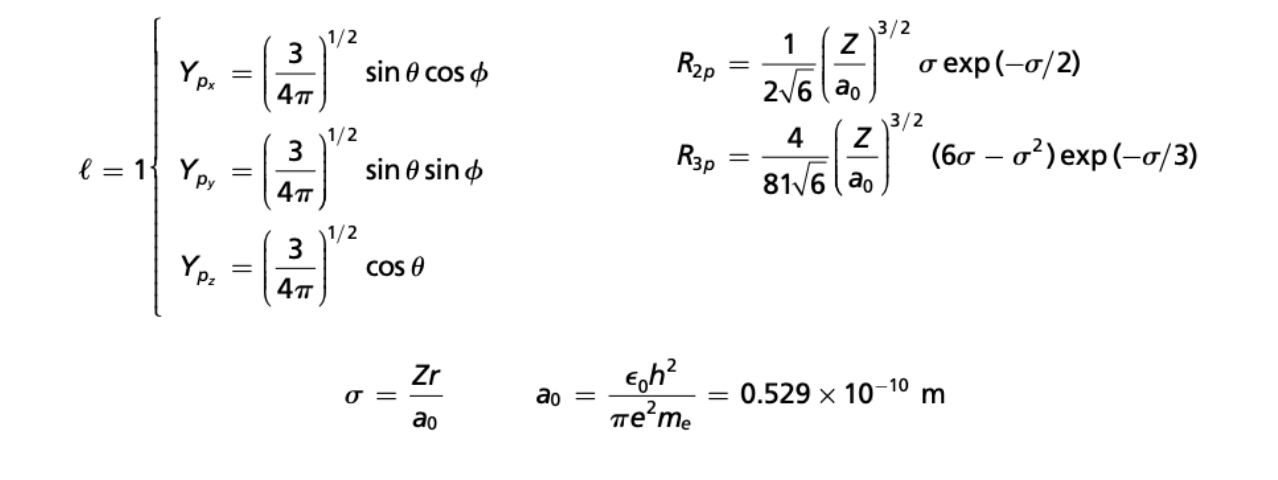
r = 6a0 = 3.174Å
Angular nodes:
theta = 0 and π,
phi = 2 and 3π2
(3 min) Determine the Ka of the unknown acid “HA” given that the Kb of SO32- is 9.80 x 10-8 and the equilibrium constant for the reaction below is 5.1 x 10-12.
HA + SO32- → HSO3- + A-
Ka = 5.00 x 10-5
(3 min) Calculate the Gibbs free energy change of the following reaction at 527ºC if the initial partial pressures of SO2, O2, and SO3 are 0.50 atm, 1.60 atm, and 1.20 atm (respectively) and the reaction has an equilibrium constant of 7.900 x 108 at 527ºC.
2 SO2 (g) + O2 (g) ⇋ 2 SO3 (g)
DeltaGrxn = -127.77 kJ/mol
(4 min) State the oxidation numbers for each atom in the below half-reactions that make up an electrochemical cell. What is the Gibbs free energy (in standard conditions) of the cell reaction if dichromate (Cr2O72-) is oxidized? Is this value concurrent with our qualitative expectations based on the reduction potentials?
Cr2O72- (aq) +14 H3O+ (aq) + 6 e- → 2 Cr3+ (aq) +21 H2O (l) Eored = 1.33 V
PbSO4 (s) + 2 e- → Pb (s) + SO42- (aq) Eored = -0.357 V
Cr6+ ; O2- ; H+ ; Cr3+ ; Pb2+ ; S6+ ; Pb0
DeltaGorxn = 976.62 kJ
Given that the species with the higher reduction potential was oxidized, we would expect that this redox reaction will be nonspontaneous. The positive change in free energy supports that qualitative expectation.
(3 mins) Salivary amylase is present in the mouth and breaks down complex carbohydrates into simpler sugars in chemical digestion. Consider the reaction of 2.00 M of a starch polymer with 5 x 10-6 M salivary amylase. Salivary amylase releases substrate at one-eighth the rate it releases product, and binds substrate at 100,000 times the rate it releases substrate. If k2 is 4 x 103 s-1, determine the rate of reaction.
2.00 x 10-2 M/s
(3 min) Calculate the entropy change from heating 1 mol N2 from 50K to room temperature. ∆Hvap = 5.58 kJ/mol, ∆Hfus = 0.360 kJ/mol, and the solid, liquid, and gas cP values are 2.00, 1.43, and 1.04 J/gK (respectively). Nitrogen melts at -210.ºC and boils at -195.8ºC. Assume constant pressure.
∆S = 138.4 J/K
(6 min) Consider an electron in a 2D box which is 16.0 nm by 12.0 nm. The electron can emit 3 distinct wavelengths. Find the location of all nodes and the maxima and minima of the wave function.
Quantum state: [1,2]
Node: y = 6 nm
Maxima: {8,3}
Minima: {8,9}
(5 min) A 400 mL solution of hypochlorous acid (HClO) is titrated with 2.00 M potassium hydroxide (KOH). If 150 mL of KOH were required to reach the equivalence point, please determine the pH of the solution when 65 mL of KOH were added. The Ka of hypochlorous acid is 3.0 x 10-8.
pH = 7.407
(5 min) The following reaction is carried out in a constant pressure calorimeter at atmospheric pressure (1 atm) and room temperature (25ºC) with 36.033 g of carbon and excess hydrogen gas. If the heat capacity of the calorimeter is 6450 J/K and the reaction temporarily heats up the calorimeter by 34.795ºC, calculate the change in internal energy and the molar enthalpy of the reaction at 25ºC:
C (s) + 2 H2 (g) → CH4 (g)
Delta Horxn = -74.81 kJ/mol
Delta U = -221.95 kJ
(5 min) A Galvanic cell is constructed according to the half-reactions and balanced redox reaction below. If the 0.500 L anode solution (at 25ºC) is initially at pH = 10 and Cu+ is added as 0.500 mol CuNO3, please determine the voltage generated by the cell.
Mn(OH)3 (s) + e- → Mn(OH)2 (s) + OH- (aq) Eored = -0.40 V
Cu+ (aq) + e- → Cu (s) Eored = 0.522 V
Mn(OH)2 (s) + OH- (aq) + Cu+ (aq) → Mn(OH)3 (s) + Cu (s)
Delta Ecell = 0.685 V
(5 min) Given the following elementary steps, prove that the rate of the overall reaction is equal to the rate indicated below. You may assume steady state conditions for all intermediates:
A + B → C
2B + C → D
C → E
D + E → F
(k_1[A][B]^2sqrt(k_2k_3))/(k_2[B]^2+k_3)
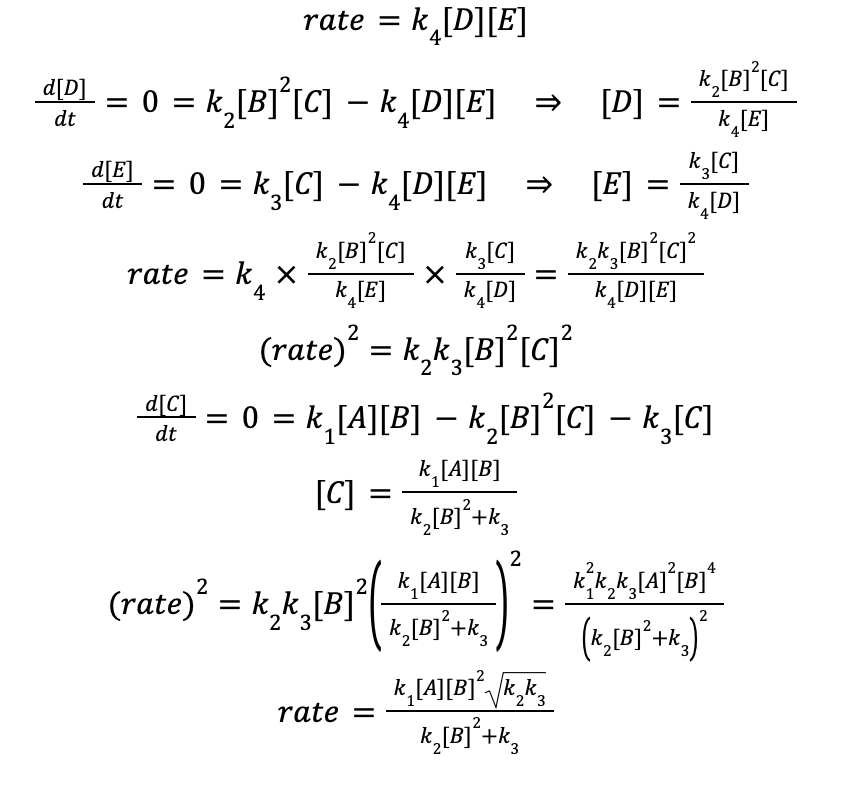
(7 mins) A galvanic cell is constructed from the following two half-reactions:
ClO4- (aq) + 2 H3O+ (aq) + 2 e- → ClO3 (aq) + 3 H2O (l) Eored = 1.19 V
I2 (s) + 2 e- → 2I- (aq) Eored = 0.535 V
The reaction is conducted in standard conditions with the exception of the concentration of H3O+. Instead of adding 1.00 M H3O+, 1.00 M of a weak acid (“HA”) is added. If the cell produces 0.312 V at these initial concentrations, please determine the Ka of the weak acid. (R = 8.314 J/Kmol, F = 96,485 C/mol)
Ka = 2.53 x 10-12
Final Jeopardy! Category: Concentration Cell.
You have 1 minute to determine your wager.
An electrolytic cell is constructed to isolate lithium metal. At the anode, hydrogen gas is bubbled at a partial pressure of 1 atm through a buffered solution past a platinum electrode. At the cathode, a platinum electrode is submerged in a 3M solution of LiCl.
The standard reduction potential of Li+ is -3.04 V.
The anode is buffered by a weak acid HA with pKa 4.75, which is prepared by addition of 3 equivalents of HA with 1 equivalent of its conjugate base A-.
To power this electrolytic cell, you may choose between four batteries of voltage 2V, 3V, 4V, or 5V. What is the minimum amount of energy required to isolate 2.0 g of lithium metal, assuming the concentrations of Li+ and H3O+ at the cathode and anode respectively do not change appreciably throughout the reaction? Assume standard temperature.