What is chemical bonding
joining of atoms to form a compound or molecule
Where are metals located on the periodic table?
left
What do the dots on in the Lewis dot structure represent?
What type of elements form ionic bonds
metals and non-metals
What type of elements form covalent bonds?
nonmetals
Why do atoms bond?
to become more stable (fill their outer shell of electrons)
Where are non-metals located on the periodic table?
right
How many valence electrons does oxygen have
6
What happens to electrons in an ionic bond
they are transferred
What happens to electrons in a covalent bond
they are shared
What determines how an atom will bond?
the number of electrons
the position of the elements on the periodic table
What type of element is oxygen?
non-metal
How many valence electrons does sodium have
1
What charge does a metal become after losing electrons
positive
Is this an example of a covalent bond? Explain why.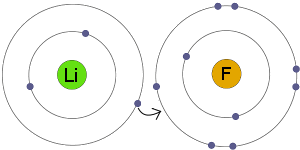
No, it is ionic. It show lithium (Li) and fluorine (F). Li is a metal and F is a non-metal.
What type of elements usually form bonds: metals, non-metals, or both?
both
What type of element is magnesium (Mg)
metal
What is the maximum number of electrons shown in a Lewis dot structure?
8
What charge does a nonmetal become after gaining electrons
negative
Name an example of a covalent bond. DO NOT USE LiF or H20!!!
answer should contain 2 nonmetals
What is a compound?
a substance made of two or more different elements chemically bonded
Is hydrogen a metal?
no, despite it being located on the left of the periodic table
Why are Lewis dot structures important?
they show how atoms will bond
What is an example of an ionic compound.
DO NOT USE NaCl!!!
answer should show a metal and nonmetal
Why do atoms share electrons in covalent bonds
to become more stable and to fill their outermost energy shell