In a Chemical Reaction these are broken or created to form new molecules.
What are bonds?
The movie we watched on Tuesday and Today?
What is Coco?
As more collisions in a system occur, there will be more combinations of molecules bouncing into each other, resulting in a higher chance that the molecules will complete the reaction and therefore the rate of that reaction will increase.
What is the Collision Theory?
The study of the amounts of substances involved in reactions.
What is Stoichiometry?
This common chemicsl reaction occurs when iron molecules react with oxygen molecules?
What is rusting?
The word most often used to start a Contexto game.
What is tree?
Temperature, energy, concentration, and pressure.
What are the factors that affect the rate of reactions?
The measurement of heat that chemists use.
What are Joules?
Finish this statement: "A reaction could include atoms compounds, molecules, or _______ of a single element.
What are ions?
The TSP teacher at Kenilworth Junior High School.
Who is Ms. Selna?
The only state of matter that changes its' reaction rates when pressure is either increased or decreased.
What is a gas?
The mass of reactants, mass of products, chemical equations, molecular weights of reactants & products, and formulas of various compounds.
What one might look at when studying Stoichiometry?
The two components that make up a reaction.
What are "products" and "reactants"?
The tallest plant in the large metal planter box outside.
What is a sunflower?
Substances that can speed up a reaction without being consumed in the process.
What are catalysts?
Represented by the variable 'K', this number is the sum of all the actions and conditions in a reaction.
What is the Equilibrium Constant?
The Type of reaction this image is an example of.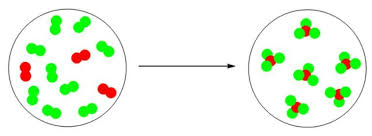
What is a synthesis reaction?
What the acronym TSP stands for.
What is the Therapeutic Support Program?
This is reached when the forward and reverse reactions occur at the same rate causing the concentrations of reactants and products to remain constant.
What is equilibrium?
When new molecules or substances are added that are not a part of the main reaction, when the temperature of the system is changed, when the pressure of the system is changed, when the concentrations are changed, and when the total volume of the system is changed.
What are the things that can change the "Equilibrium Point" during a reaction?