How would you write the chemical formula for this molecule?
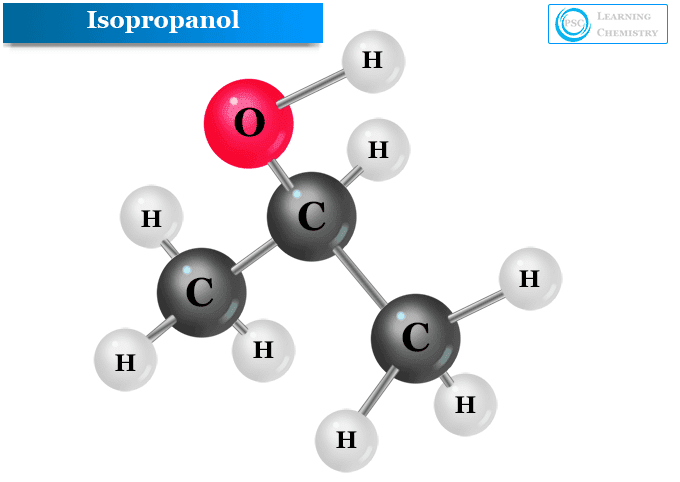
C3H8O
(isopropanol or rubbing alcohol)
A change in __________, __________, or __________ are all signs of a chemical change. (Must get all 3!)
color, temperature, smell
What does it mean for a chemical equation to be "balanced"?
There is the same type and number of atoms in the reactants and the products
Which molecules are the products? Give the names.
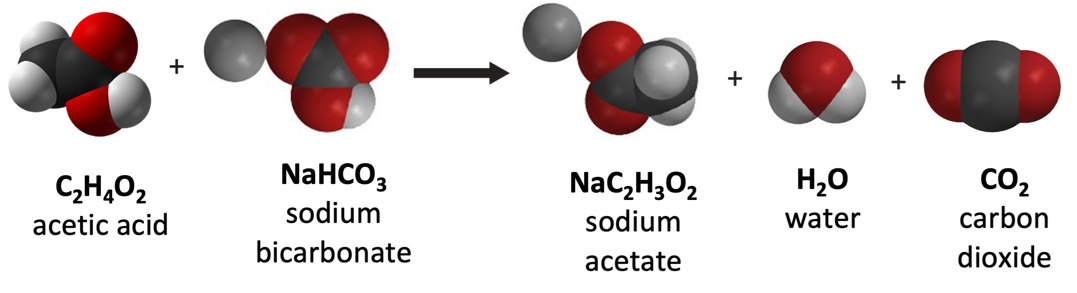
sodium acetate, water, and carbon dioxide
For their volcano experiment at the science fair, 3 students mixed baking soda and vinegar. The mixture immediately started fizzing and bubbling out of the volcano. Do you think a chemical reaction happened? Why or why not?
:max_bytes(150000):strip_icc()/volcano-experiment-175499267-5c3a0160c9e77c000176058c.jpg)
Yes, a gas was formed (bubbling and fizzing).
How would you write the chemical formula for this molecule?
(black=carbon, white=hydrogen, red=oxygen)
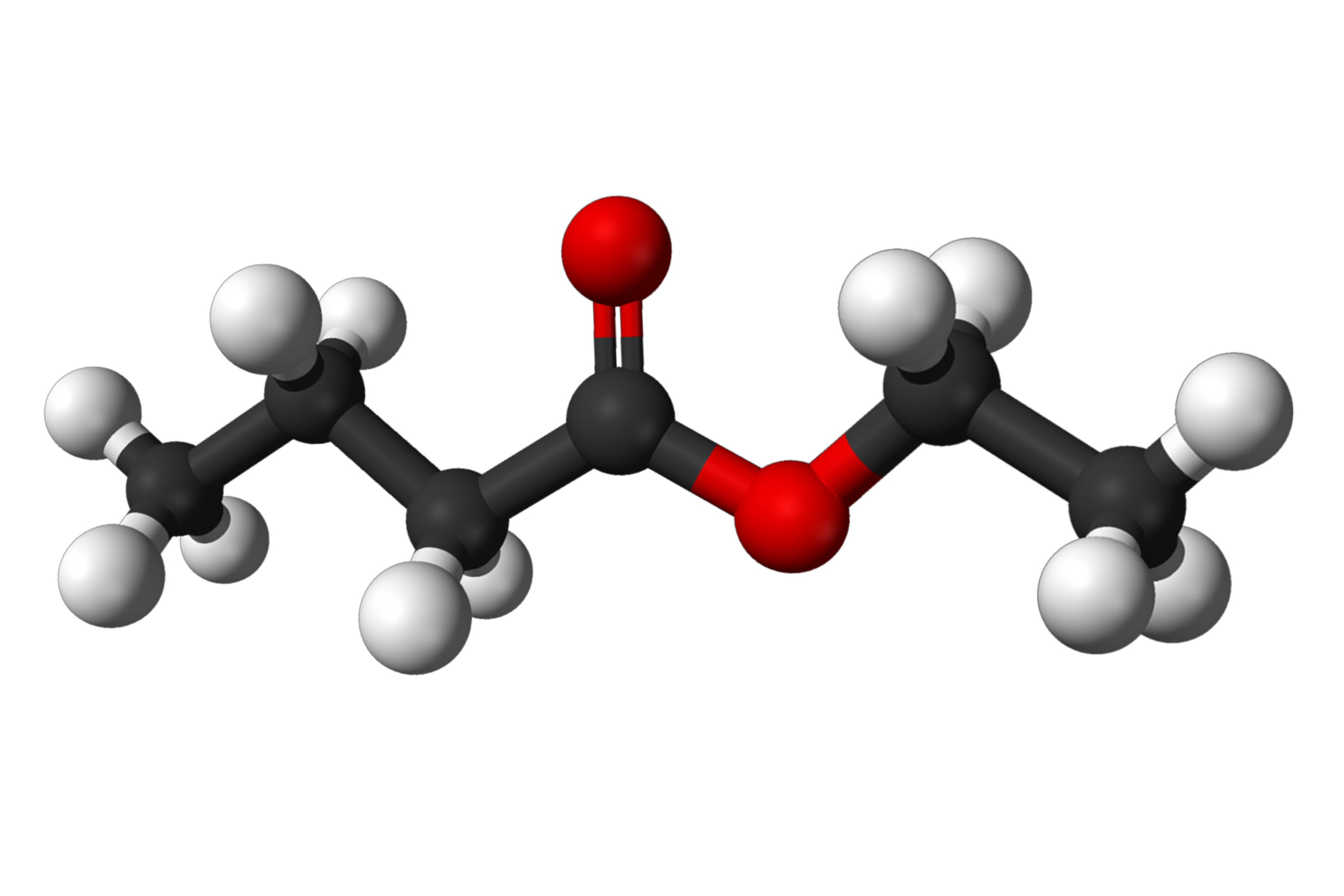
C6H12O2
(ethyl butyrate)
Give 3 examples of physical changes.
Lots of answers: slicing an apple, boiling or freezing water, crumpling paper, breaking a pencil, etc.
Is this chemical equation balanced? If yes, how many of each atom are on each side? If not, what other atoms does it need, and on which side, to become balanced?
(grey=carbon, red=oxygen)

No, there should be 1 more oxygen in the products.
Which molecules are the reactants? Give the names.
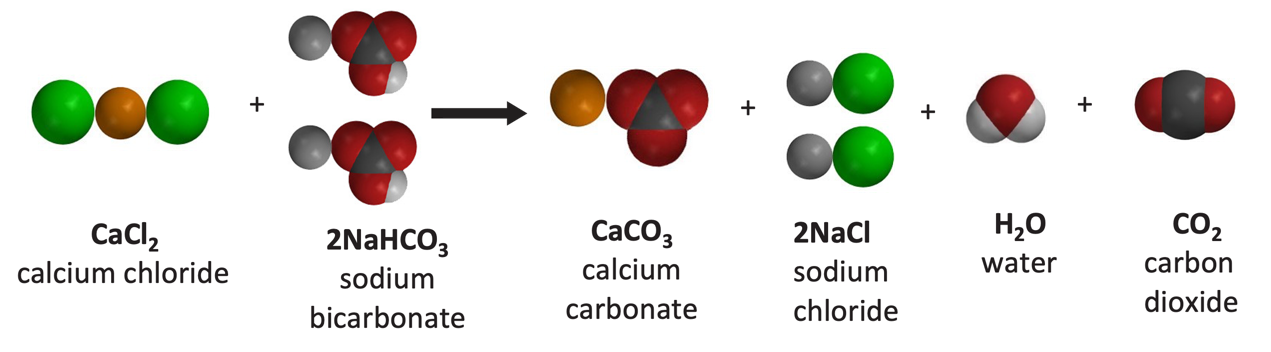
calcium chloride and sodium bicarbonate
A student mixed together 2 odorless substances at room temperature. The mixture immediately became very cold and smelled like old cheese. Do you think a chemical reaction happened? Why or why not?
Yes, there was a change in temperature AND a change in smell.
Which elements + how many of each are in a CH3COOH molecule? (acetic acid)
![]()
2 carbons, 4 hydrogens, and 2 oxygens
What is the main difference between chemical and physical changes?
Chemical: A new substance/molecule is formed
Physical: No new molecules are formed, just a change in appearance
Is this chemical equation balanced? If yes, how many of each atom are on each side? If not, what other atoms does it need, and on which side, to become balanced?
(grey=carbon, white=hydrogen, red=oxygen)
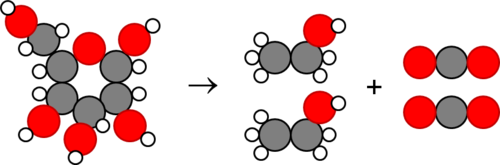
Yes, there are 6 carbons, 12 hydrogens, and 6 oxygens on each side.
Fill in the blank: Jack represented a benzene _________ using clay and toothpicks.

molecule
A student mixed a green liquid with a blue liquid. The mixture turned orange and the texture became thicker. Do you think a chemical reaction happened? Why or why not?
Yes, there was an unexpected color change and a precipitate (a new solid) was formed.
Which elements + how many of each (total) are in 2 H2SO4 molecules? (sulfuric acid)
![]()
4 hydrogens, 2 sulfurs, and 8 oxygens
List the 5 common signs that a chemical reaction has occurred. (Must get all 5!)
Change in color, change in temperature, change in smell, formation of a gas (bubbling/fizzing), formation of a solid (precipitate)
Is this chemical equation for photosynthesis balanced? If yes, how many of each atom are on each side? If not, what other atoms does it need, and on which side, to become balanced?
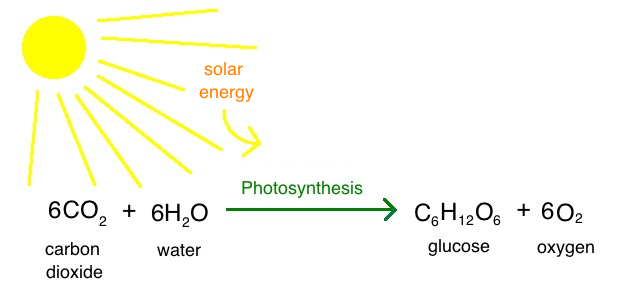
Yes, there are 6 carbons, 12 hydrogens, and 18 oxygens on each side.
What is the definition of a "chemical reaction"? (paraphrasing is okay)
A process in which atoms rearrange to form new molecules
A student put a melted Otter Pop in the freezer. An hour later, the Otter Pop had frozen. Do you think a chemical reaction happened? Why or why not?
No, a liquid freezing into a solid is a physical change. No new molecules were made.
Which elements + how many of each (total) are in 3 C8H10N4O2 molecules? (caffeine)
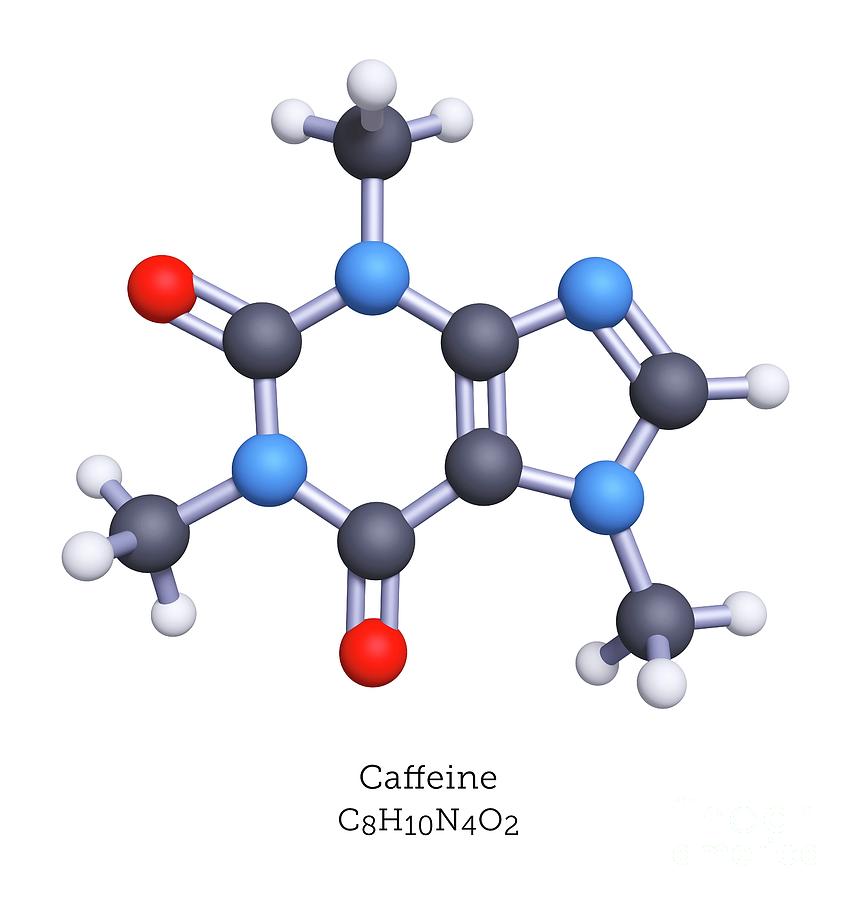
24 carbons, 30 hydrogens, 12 nitrogens, and 6 oxygens
Explain why boiling water into steam is a physical change.
Steam is just water in its gas form. Steam and liquid water are both made of H2O molecules, so no new molecules are formed.
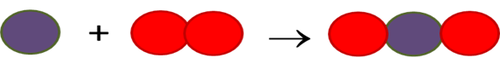
Is this chemical equation balanced? If yes, how many of each atom are on each side? If not, what other molecules does it need, and on which side, to become balanced?
(white molecule=H2, red molecule=O2, product molecule=H2O)
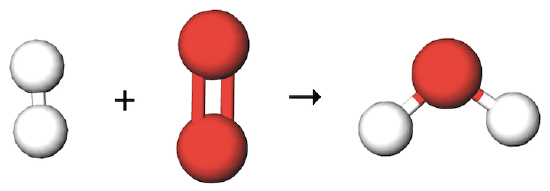
No, there should be 1 more H2 molecule in the reactants and 1 more H2O molecule in the products.
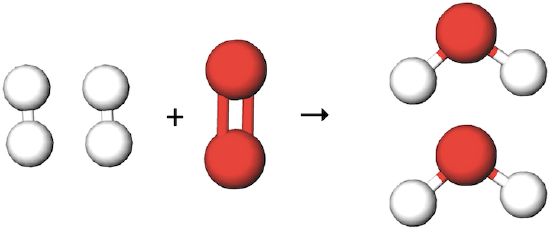
Give the definition of "property" AND give 3 examples. (paraphrasing is okay)
A characteristic of a substance that can be observed or measured
Examples: color, smell, boiling point, melting point, texture, taste, etc.
A student mixed a blue liquid with a yellow liquid. The mixture turned green (but nothing else happened). Do you think a chemical reaction happened? Why or why not?
No, because blue + yellow = green. It turned the color we would expect, and no other signs of a chemical reaction happened.