Positively charged ions are deficient in this subatomic particle.
What are electrons?
This bond is the transfer of electrons between a nonmetal and a metal.
What is an ionic bond?
The product(s) of the following reaction: C3H8 + O2.
What is entropy?
The suffix for fully saturated hydrocarbons, i.e., CnH2n+2.
What is -ane?
This is the name given to metals in the second group of the periodic table.
What are alkali-earth metals?
This bond is the unequal sharing of electrons between two nonmetals.
What is a polar covalent bond?
The precipitate product of the following reaction: NaCl + AgNO3.
What is AgCl?
The term for the energy "barrier" that molecules need to overcome in a collision for a reaction to occur.
What is activation energy?
The hybridization of carbon in ethene (CH2CH2).
What is sp2?
This element has the largest radius of any known element.
What is Francium?
The melting and boiling points of the halogens increase going down their group primarily due to this IMF becoming stronger.
What are LDFs?
The product(s) of the following reaction: CaCO3 + heat.
The relationship between the change in Gibbs free energy and the equilibrium constant of a reaction.
The IUPAC name of CH3CH2COOH.
What is propanoic acid?
This is the noble gas electron configuration for Copper.
What is [Ar] 4s1 3d10
These are the strongest types of intermolecular forces between molecules of CHF3.
What are dipole-dipole forces?
The product(s) of the following reaction: Li + N2.
What is Li3N?
An endothermic reaction that decreases in entropy will be spontaneous under these conditions.
What is never?
The number of chiral centers in the pictured molecule.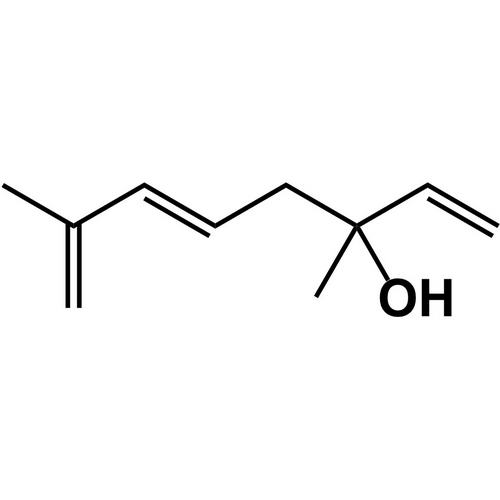
What is 1?
This element has the highest electron affinity of any element.
What is Chlorine?
This is the molecular geometry of XeF4.
The product(s) of the following reaction: MnO4- + H+ + H2O2.
What are Mn2+, O2, and H2O?
This is the relationship between work w, heat q, and change in internal energy dU of a reaction.
What is dU = w + q?
The mechanism by which fluorethane (CH3CH2F) will react with sodium hydroxide (NaOH).
What is SN2?