How many neutrons are in Boron-11
What is 6 neutrons?
The number of electrons that can exist in one orbital
What is 2?
The number of significant figures in 0.084930
What is 5?
Two or more elements in a fixed mass ratio.
What is a compound?
The density of a metal cube with a volume of 20 cm3 and a mass of 12 g.
What is 0.6 g/cm3?
How many protons, neutrons, and electrons?
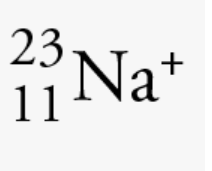
protons: 11
neutrons: 12
electrons: 10
As wavelength increases, the frequency _______
What is decreases.
The number of significant figures in 9,076,400
What is 5?
Determines the atomic number of an element.
What is the number of protons?
What is the volume of 20 grams of liquid that has a density of 100 g/ml?
0.2 ml
What are the masses of these isotopes?
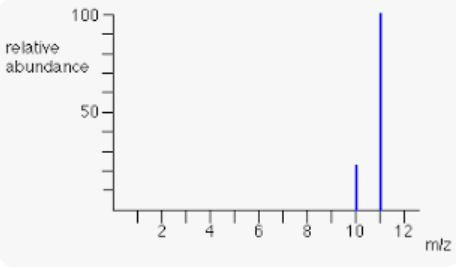
What is 10 and 11 g/mol?
The number of electrons that can fit into the 4th energy level.
What is 32?
The answer of 8.76 / 9.065 with correct significant figures.
What is 0.966?
A substance that is not broken down by physical or chemical means.
What is an element?
The density of aluminum is 2.7 g/ml. A sample of 25 cm2 of metal has a mass of 100 grams. Is the sample aluminum?
100/ 25 = 4 g/cm2, so no
Which is not a pair of isotopes?
3216 S, 3316 S
126C, 146C
168O, 167O
168O, 167O
Responsible for energy in nuclear reactors, involving the splitting of heavy atoms.
What is fission?
Name the trend: The greatest values are at the bottom and to the left.
What is atomic radius?
The scientist that developed the orbital model of the atom with discrete levels
Niels Bohr
What fraction of a substance will remain after five half lives?
1/32
What is the average molar mass of this element?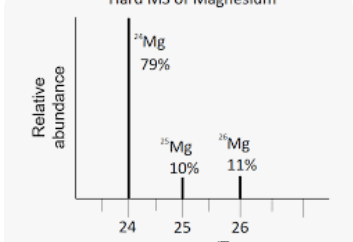
What is 24.32 g/mol
The element with the electron configuration of 1s22s22p63s23p4
What is sulfur?
What region on the periodic table is ionic radius the greatest?
What is to the right and near the top?
Developed the first periodic table.
Who is Dmitri Mendeleev?
The half-life of Zn-71 is 2.4 minutes. If one had 100.0 g at the beginning, how many grams would be left after 7.2 minutes has elapsed?
What is 12.5 g?
What element is this? 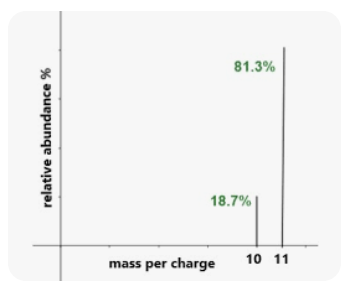
What is Boron?
(0.813 x 11) + (0.187 x 10)= 10.8
Radioactive particles and degree of harm.
What is alpha- least harmful
Beta-low to moderate harm
Gamma-very harmful.
A group is reactive with a valence electron of one.
What are alkali metals?
Developed the plum pudding model with his discovery of electrons using cathode ray tubes.
Who is JJ Thompson?
How long will it take for 18.0 grams of Ra-226 to decay to leave a total of 2.25 grams? Ra-226 has a half-life of 1600 years.
What is 4800 years?