If the value of Keq is lower than one, the reaction is favored where?
Reactant
In an endothermic reaction, what happens to the equilibrium when you increase the temperature?
Shifts toward the products
According to the common ion effect, what would precipitate out of a solution?
The less soluble salt
What is(are) the only state(s) of matter that does affect a reaction's equilibrium constant?
Gases and aqueous solutions
What happens when you remove copper nitrate from this reaction?
HCl (aq) + Cu(NO3) (aq) ⇆ CuCl (s)+ HNO3 (aq)
What could you add to a reaction that would not change the equilibrium of a reaction, but only the speed of the reaction?
Catalyst
What is Le Châtelier’s Principle?
When a stress is placed on a reversible process, it will proceed in the direction that relieves the stress
What is the solubility of BaSO4? (Ksp = 1.1 * 10-10)
2.4 * 10-3 M
Prove that a 2.1 * 10-7 M Ag3PO4 solution is either saturated or unsaturated.
Unsaturated
Its ion product concentration (5.25 * 10-26) is less than its accepted Ksp of 1.5 * 10-19.
What happens when you lower the pressure of the following reaction?
N2 (g) + 3H2 (g) ⇆ 2NH3
Equilibrium will shift toward the reactant
For the following reaction, indicate the value and formula of the equilibrium constant and indicate if it is product- or reactant-favored.
2AlCl3 (s) → 2Al (s) + 3Cl2 (g)
Keq = 2.74
Keq = [Cl2]3
Product-favored
For the following reaction, indicate the value and formula of the equilibrium constant and indicate if it is product- or reactant-favored. The concentration of magnesium chloride is 3.7 M, of magnesium hydroxide is 5.7 M, and of hydrochloric acid is 2.3 M.
Mg(OH)2 (aq) + 2HCl (aq) → MgCl2 (aq) + 2H2O (l)
Keq = .123
Keq =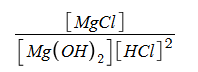
Reactant-favored
What is the Haber process? Why was it hard to make a lot of ammonia? What conditions made the Haber process possible?
Used to produce large amounts of ammonia for fertilizers, cleaning compounds, and explosives
Ammonia was hard to mass-produce because the high Keq favors the products BUT it takes forever to reach equilibrium.
Conditions for the Haber process include high pressure, high temperature (in addition to a catalyst to speed up the reaction), and removing product so that the equilibrium favors the product side
Would a precipitate form if the same amount of 5.93 * 10-5 M Pb(NO3)2 and 1.5 * 10-6 M Na3PO4? If so, what is it?
Yes
Pb3(PO4)2
Ion product concentration (1.2 * 10-25) is higher than the accepted Ksp
Will a precipitate form if the same volume of 5.3 * 10-6 M of Ba(NO3)2 and 2.25 * 10-5 M of Na2CO3? If so, what would it be?
No, because ion concentration is 3.0 * 10-11 (less than the Ksp of BaCO3)