What are compounds with the same molecular formula but different structural formulas?
Isomers
Draw the following hydrocarbon and indicate what family it is part of:
Pentanal
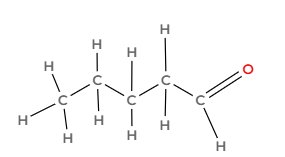
Aldehyde
Name the following hydrocarbon and state what family it belongs to.

Iodine haloaromatic ring
Family: haloraomatic
What is a Carbonyl group?
C=O
Are short or long fatty acids solid at room temp?
Long fatty acids
Draw the following hydrocarbon and indicate what family it is part of:
Pentoxyheptane
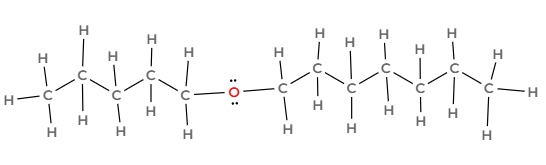
Ethers
Name the following hydrocarbon and state what family it belongs to.

Methanoic acid
Family: carboxylic acid
What is the most common aromatic hydrocarbon?
Benzene ring
What is an ether?
Hydrocarbon with an oxygen bridge between two carbon chains
Draw the following hydrocarbon and indicate what family it is part of:
2-methylbutanol

Alcohol
Name the following hydrocarbon and state what family it belongs to.

2-methylnonanamine
Family: amine
What is an alkyl group?
An additional hydrocarbon side chain on an alkane with a hydrogen missing (so it can bind to a larger molecule)
Aldehydes - organic compounds that contain an oxygen held with a double bond to an end carbon
Ketone - when a carbonyl group is located within a carbon chain (not on the end)
Draw the following hydrocarbon and indicate what family it is part of:
hexane-4-one
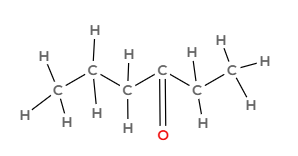
Ketone
Name the following hydrocarbon and state what family it is in.
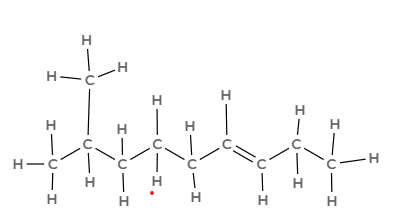
2-methylnon-6-ene
Alkene
What is an addition reaction?
Occurs when a multiple bond of a molecule is broken and two atoms or groups of atoms are added.
Give three reasons explaining why carbon is in every organic compound.
Carbon has four valence electrons
Carbon can form single, double, or triple bonds
Carbon forms stable and nonpolar bonds with hydrogen
Draw the following hydrocarbon and indicate what family it is part of:
butyl pentanoate

Ester
Name the following hydrocarbon and state what family it is in.

4-ethyl,2,5-methyloctane
Alkane
Between aromatic compounds and aliphatic compounds, which one has delocalized electrons?
Aromatic compounds