The arrangement of electrons in an atom.
What is electron configuration?
Fluorine, chlorine, bromine, iodine, and astatine.
What are the halogens?
The number of energy levels for hydrogen and helium.
What is one?
This model states that all particles are in constant motion and the temperature of the substance determines the speed of the molecules.
What is the Kinetic Molecular Theory?
The ground state electron configuration of nitrogen (N).
What is 1s2 2s2 2p3?
The change of a solid directly to a gas.
What is sublimation?
Helium, neon, argon, krypton, xenon, and radon.
What are the noble gases?
The number of electrons that could be found in the 3rd energy levels.
What is 18?
This model states that it is impossible to know the position and the velocity of a subatomic particle at the same time.
What is the Heisenberg Uncertainty Principle?
This is the orbital notation for oxygen.
What is: 
The measure of the average kinetic energy of particles in matter.
What is temperature?
Hydrogen, lithium, sodium, potassium, rubidium, cesium, and francium.
What are the alkali metals?
The outer energy level.
What is the valence shell?
This theory explains chemical bonding as the overlap of atomic orbitals creating a shared electron pair.
What is the Valence Bond Theory?
The number of protons, neutrons, and electrons for manganese (Mn).
What is 25, 55, and 25?
This shows that any sample of carbon dioxide always contains 12 grams of carbon to 32 g of oxygen.
What is the law of definite composition?
Nitrogen, phosphorous, arsenic, antimony, bismuth, and moscovium.
What is the nitrogen group?
The number of energy levels for sodium, magnesium, aluminum, silicon, phosphorous, sulfur, chlorine, and argon.
What is 3?
This was the first attempt to describe all matter in terms of atoms.
What is Dalton's Atomic Theory?
The number of total atoms in 3 molecules of Ca3(PO4)2.
What is 39?
Any property that can be observed or measured without altering its chemical composition.
What are physical properties?
Boron, silicon, germanium, arsenic, antimony, and tellurium
What are the metalloids?
The number of valence electrons for family 14.
What is 4?
This model explains chemical bonding by combining atomic orbitals into new molecular orbitals.
What is the Molecular Orbital Theory?
The electron dot notation for the covalent compound CO2.
What is: 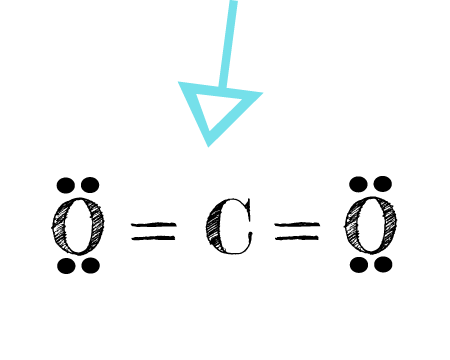 ?
?