Vegetable oil is what kind of substance?
Lipid/Fat
An example of a physical change
not changing the chemical properties it has
What is the ranges of bases?
8-14
The number one solvent...
Water
When animals eat food containing protein they consequently obtain....
Amino Acids/Proteins
Why are fats important nutrients?
Because they are able to store energy
Water changed from a liquid to a gas...
Physical Change
What is the ranges of acids?
1-6
How do we determine if something is acidic or basic?
Using Lithmus Paper
Starch molecules are broken into smaller units known as...
simple sugars
Building blocks of proteins
Amino Acids
What event must always occur for a chemical reaction to take place?
effective collisions between reacting particles
Compounds/elements chemically combining
Which molecule would fit into the enzyme?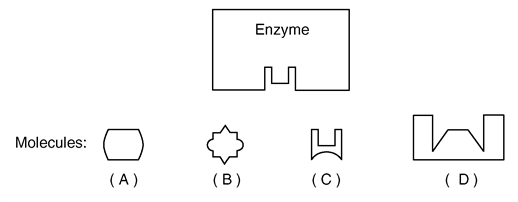
Molecule C
How is a base different from an acid?
Contains more OH– ions than H+ ions
All enzymes are considered these...
Proteins
The long chain of lipids that are considered the building blocks.
Fatty acid
The process of boiling is considered to be what kind of change and why?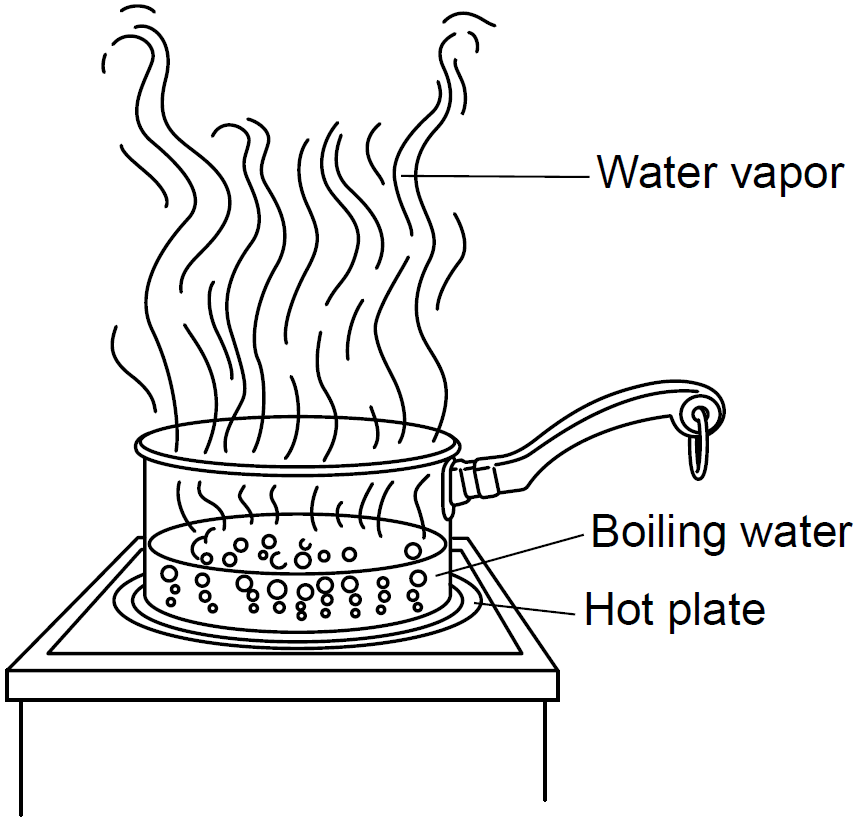
Physical change because it is NOT creating a new substance.
If something has a pH level of 7 that would make the solution what?
Neutral
What kind of particles do Acids have?
H+ ions
What does this formula represent and what kind of macromolecule is it?
C6H12O6
Formula - Glucose
Carbohydrate
The three major components that make up a nucleic acid (NOT THE ELEMENTS!)
nitrogenous base, phosphate group, & 5-carbon sugar
The diagram shows a chemical equation with the substances on either side of the equation hidden from view. From the law of conservation of mass, we know that..
The reactants and the products will have the same mass
The substance that gets dissolved in a solution
A solute
When a teaspoonful of sugar is added to water in a beaker, the sugar dissolves. The resulting mixture is a what?
Solution