What is an atomic number on the periodic table?
The number in the top left of the box. Shows the number of protons in an element.
How many electrons does an element want to have for a full outer shell?
Eight (8) electrons
Which element is more electronegative: Sodium (Na) or Potassium (K)?
Does Neon (Ne) have a strong electronegativity if it has a full outer shell of electrons?
No - it has no electronegativity
What is cohesion?
Intermolecular attraction between the same substance
What are intermolecular forces?
The attractive / repulsive forces acting between atoms.
How many valence electrons does Oxygen have?
Six (6) electrons
Why does Carbon (C) have a higher electronegativity than Boron (B)?
Because it has more electrons making a stronger attractive force
Ionic
What is a covalent bond?
An electron being shared between two atoms, creating a bond between two atoms.
What are small, basic molecules that act as building blocks for larger compounds?
Monomer
What charge do neutrons have?
Neutral charge
Which element is more electronegative: Nitrogen (N) or Oxygen (O)
Oxygen
*DAILY DOUBLE*
What type of bond is created?
Ionic Bond
What is the gravity-defying flow of liquid upward through narrow spaces called?
Capillary Action
What are valence electrons?
Electrons on the outermost shell on an element
How many protons does Potassium (K) have?
19 Protons
Why does Chlorine (Cl) have a higher electronegativity than Argon (Ar)?
Argon has a full outer shell meaning it does not want to gain or lose any more electrons. This means it also has the weakest attractive force
What type of bond is formed with Carbon Monoxide (CO)?
Covalent
What is an ionic bond?
An electron being taken by another atom, creating a bond between two atoms.
What is an intermolecular force?
The attractive or repulsive force between molecules / atoms
*DAILY DOUBLE*
How many TOTAL electrons does Bromine (Br) have?
35 total electrons
*DAILY DOUBLE*
Why does Oxygen have a higher electronegativity than Sulfur?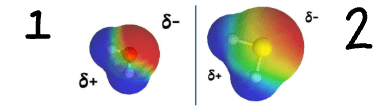
Because Oxygen has a smaller atomic radius
What type of bond is formed with Sodium Phosphate (Na3P)?
Ionic
What is a large molecule made of repeating structures?
Polymer