In the hypothetico-deductive method, what comes right after making observations?
A) Testing
B) Hypothesizing
C) Analyzing data
D) Drawing conclusions
B) Hypothesizing
The smallest unit of matter that still retains the properties of an element is a(n):
A) Proton
B) Atom
C) Neutron
D) Molecule
B) Atom
What type of bond connects two water molecules?
Hydrogen bond
What is the concentration of hydroxide ions in a solution where pH = 10?
A) 1 × 10–10 M
B) 1 × 1010 M
C) 1 × 104 M
D) 1 × 10–4 M
D) 1 × 10–4 M
How many covalent bonds can carbon form up to?
4
Which of the following best describes a hypothesis?
A) A proven fact
B) A random guess
C) A plausible explanation that can be tested
DD A final conclusion
C) A plausible explanation that can be tested
When two atoms share one or more pairs of electrons, what type of bond is formed?
Extra 100 points : Name one molecule that forms this type of bond.
Covalent bond
Which property of water explains why lakes don’t freeze solid from the bottom up?
A) Adhesion
B) Cohesion
C) Expansion upon freezing
D) High surface tension
C) Expansion upon freezing
The hydrogen ion concentration [H+] of a solution is 1×10^5 M. What is the pH of this solution, and is it acidic, neutral, or basic?
pH 5 ; acidic
Ice floats on liquid water because of the way hydrogen bonds form when water freezes. Why is this important for life on Earth?
It keeps lakes and oceans from freezing solid, allowing organisms to survive beneath the ice
Which statement about correlation is TRUE?
A) Correlation always means causation
B) Correlation measures cause-and-effect
C) Correlation shows a relationship between variables
D) Correlation proves one variable causes another
C) Correlation shows a relationship between variables
What are the three main subatomic particles, which charge does each posses, and how are they organized within the structure of an atom?
Protons(+), neutrons(=), and electrons(-).
Protons and neutrons are in the nucleus while electrons orbit around the nucleus in electron cloud.
Name all of water properties mentioned in class
Cohesion, adhesion, high specific heat, universal solvent, less dense when frozen
When a hydroxide base is added to a solution, what happens to the concentration of OH- and pH?
The concentration of OH- and pH increase🆙🔝
If you were working with a protein that needed a certain pH to work, what would you need in the solution containing the protein?
A) salt
B) acid
C) buffer
D) carbon dioxide
C) buffer
In an experiment where plants are given different amounts of sunlight and their height is measured after 3 weeks, identify the independent variable and the dependent variable.
Independent variable: Amount of sunlight Dependent variable: Plant height (growth)
Which one of the following atoms would be most likely to form an anion with a charge of –2? AND WHY
A)
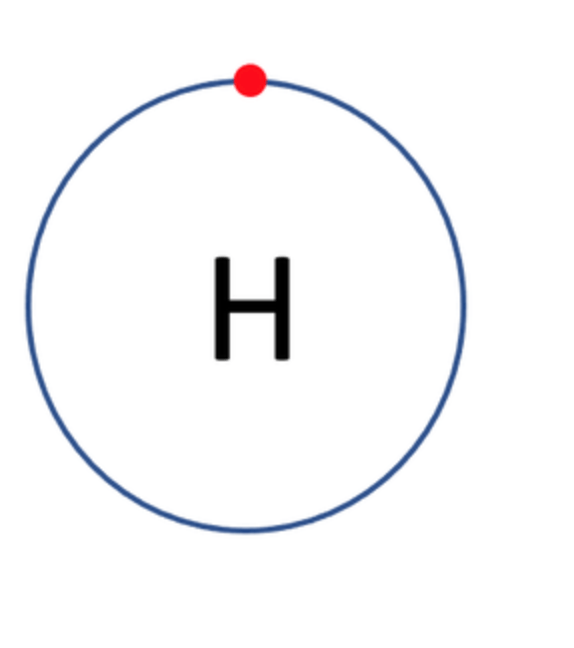
B)
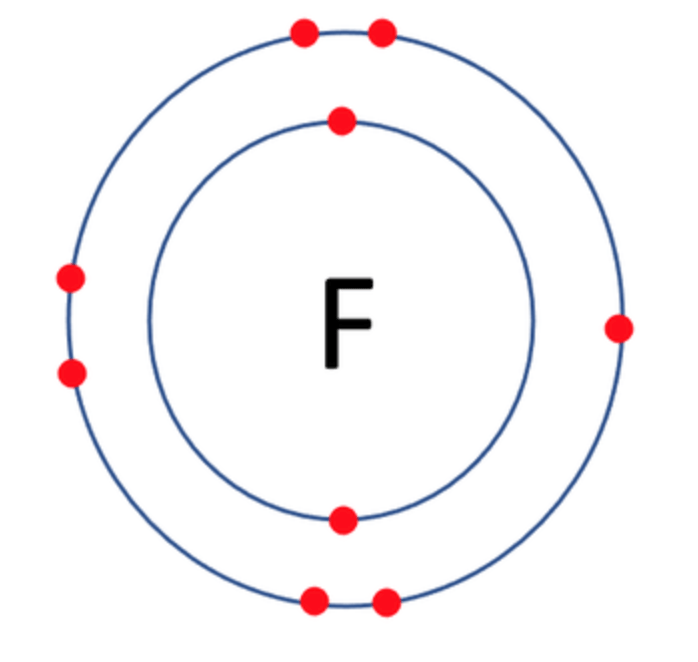 C)
C)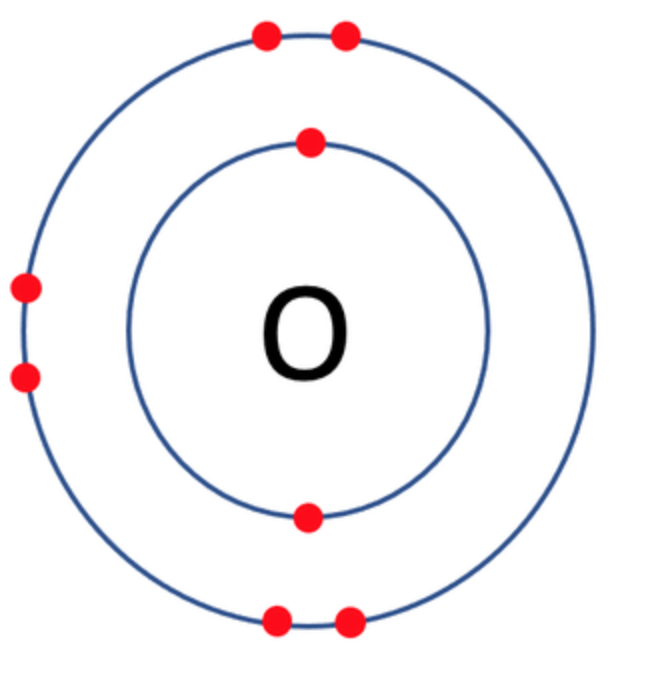
D)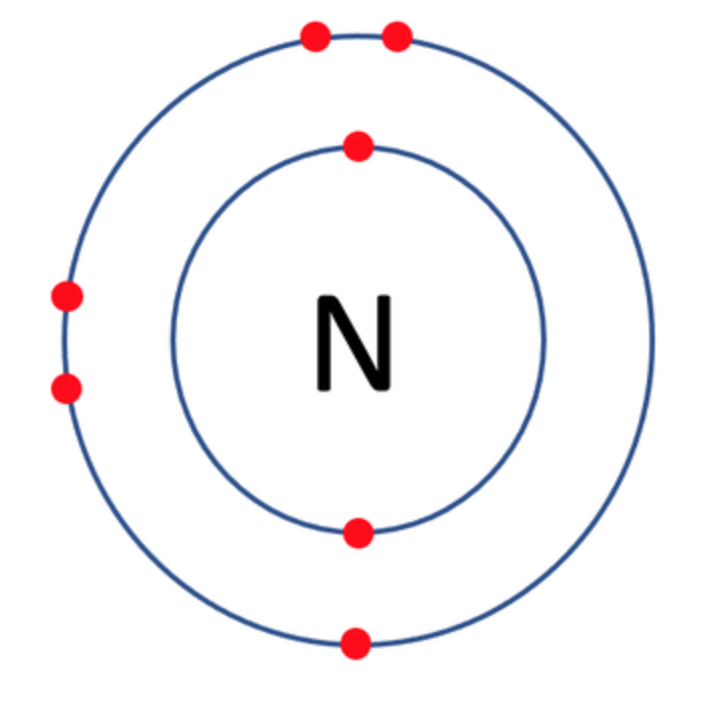
C)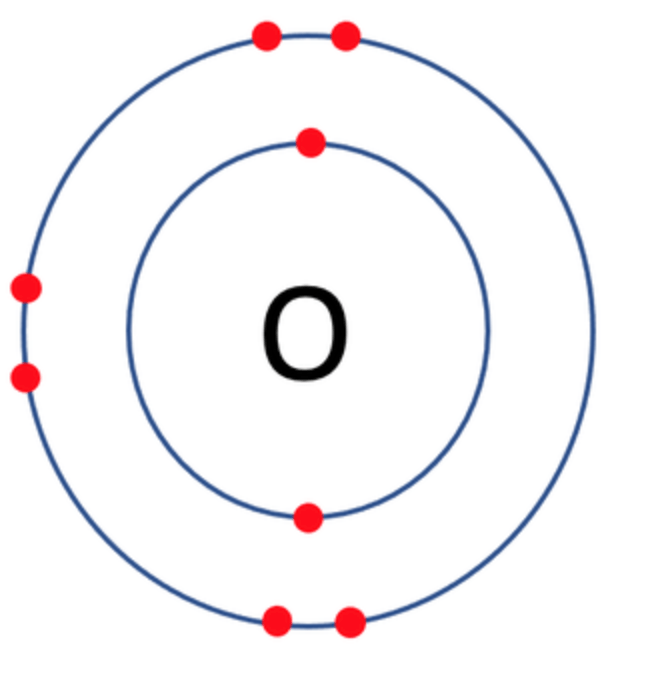
A plant relies on both cohesion and adhesion to move water upward. Which is the best explanation?
A) Cohesion holds water molecules together, while adhesion helps water stick to cell walls
B) Cohesion pushes water into leaves, adhesion prevents evaporation
C) Cohesion helps dissolve minerals, adhesion makes water hydrophobic
D) Cohesion lowers pH, adhesion raises pH
A) Cohesion holds water molecules together, while adhesion helps water stick to cell walls
To neutralize a given volume of household bleach, you would need a similar volume of what solution? (picture will be provided)
A) wine
B) battery acid
C) tomato juice
D) beer
B) battery acid
When two oxygen atoms bond together, which type of bond forms, and how many pairs of electrons are shared?
A) Single covalent bond; 1 pair shared
B) Double covalent bond; 2 pairs shared
C) Ionic bond; 2 electrons transferred
D) Hydrogen bond; 2 pairs of electrons attracted
B) Double covalent bond; 2 pairs shared
Put the statements in the right order of scientific method.
1. Two groups of plants are grown with different amounts of sunlight and measured after 3 weeks.
2. Plants with more sunlight grew taller, supporting the hypothesis.
3. Plants that receive more sunlight grow better than plants that receive less sunlight.
4. Plants near the window grow taller than plants farther away.
5.If plants receive more sunlight, they will grow taller.
4. Observe --> 3. Hypothesize --> 4. Predict --> 1. Test --> 2. Analyze and Conclude
Briefly explain what is happening to this molecule and what does this process call?

Hydrolysis is when water is added to break down the molecules
Why coastal areas have milder climates than inland areas? Explain your reasoning.
Water absorbs and releases large amounts of heat with only slight temperature change.
Absorb the heat when it's hot, releasing when it's cold
As [H]+ increases, the pH __________.
As [OH]- decreases, the pH __________.
As [H]+ decreases, the pH __________.
As [H]+ decreases, the [OH]- _________.
Extra 100 points: What is the pH range of arterial blood?
As [H]+ increases, the pH decreases
As [OH]- decreases, the pH decreases
As [H]+ decreases, the pH increases
As [H]+ decreases, the [OH]- increases
pH = 7.35-7.45
I think you got it