0.250 mol of elemental bromine, Br2, has a mass of approximately
a. 0.250 g.
b. 8.75 g.
c. 20.0 g.
d. 40.0 g.
e. 79.9 g.
40.0g
How many valence electrons are there in the correctly drawn Lewis Structure of SO32-?
26
The ion, NH4+, is called
ammonium ion.
Which of the following is best described as a covalently bonded molecule?
a) NaF
b) Cs2O
c) CaCl2
d) CS2
e) NaCl
d) CS2
The formula weight of calcium bisulfate, Ca(HSO4)2 is about
a. 69 g/mol.
b. 137 g/mol.
c. 201 g/mol.
d. 234 g/mol.
e. 274 g/mol.
d. 234 g/mol.
How many atoms are in 4.2 × 10-5 moles NH3 gas?
a. 6.0 × 1023
b. 1.2 × 1023
c. 1.0 × 1020
d. 2.4 × 1024
e. 1.0 × 10-5
c. 1.0 × 1020
How many valence electrons are in the Lewis structure of ClO3-?
26
Which of the following anions is always soluble in water regardless of its associated cation?
a. NO3-
b. S2-
c. SO42-
d. OH-
e. Cl-
a. NO3-
Correct name for SF6
Sulfur Hexafluoride
HCl (aq) + 2 Al (s) → 2 AlCl3 (aq) + 3 H2 (g)
What best describes this reaction?
a. It is a neutralization reaction.
b. It is a precipitation reaction.
c. It is an oxidation-reduction reaction.
d. It is a net ionic reaction.
e. It is an example of the combustion of albumin.
c. It is an oxidation-reduction reaction.
CH3CH2CH3 + O2 → CO2 + H2O
When correctly balanced, the coefficients of propane and carbon dioxide are respectively
a. 1 and 2
b. 1 and 3
c. 2 and 5/2
d. 2 and 4
e. 2 and 9/2
b. 1 and 3
Which molecule is bent with a Lewis structure that includes 2 lone pairs in the central atom? (Select all that apply)
a) SiCl4
b) NH3
c) H2S
d) PO2-
e) SeCl2
c) H2S
e) SeCl2
Which of the following is always soluble in water regardless of its associated anion?
a. Sr2+
b. Rb+
c. Ba2+
d. Fe2+
e Fe3+
b. Rb+
Correct name for AsF5
Arsenic Pentafluoride
Which of the following will react in a neutralization reaction with sodium hydroxide to produce
sodium nitrate?
a. sulfuric acid
b. nitric acid
c. hydrochloric acid
d. sodium sulfate
e. barium nitrate
b. nitric acid
2AuCl3 + 3Sn → 3SnCl2 + 2Au
How many moles of tin (II) chloride are produced when 3 moles of gold (III) chloride react with
plenty of tin metal present?
a. 2.0 mol
b. 3.0 mol
c. 4.5 mol
d. 6.0 mol
e. 9.0 mol
c. 4.5 mol
Which molecule is trigonal planar with a Lewis structure that includes 9 lone pairs?
a) CH3F
b) NO3-
c) SBr2
d) BI3
e) NCl3
d) BI3
The combination of ions most likely to produce a precipitate is
a. Li+ and phosphate ion
b. Pb2+ and nitrate ion
c. copper (II) ion and Cl-
d. ammonium ion and Br-
e. iron (III) and CO32-
iron (III) and CO32-
What is the correct molecular compound for dinitrogen trioxide?
N2O3
BaCl2 can be formed in a neutralization reaction solely from which pair of compounds?
a. Ba(NO3)3 and Ba(OH)2
b. Ba(NO3)2 and CaCl2
c. Ba(OH)2 and HCl
d. Ba(NO3)2 and HCl
e. NH4Cl and NH4OH
c. Ba(OH)2 and HCl
6 HCl (aq) + 2 Al (s) → 2 AlCl3 (aq) + 3 H2 (g)
About how many moles of aluminum chloride would be formed from complete reaction of 54 grams
of aluminum metal?
2.0 Moles
Which molecule is bent and has a dipole moment?
a) SiO2
b) BBr3
c) OF2
d) CH3Br
e) PBr3
c) OF2
What happens when aqueous solutions of silver nitrate, sodium sulfate, and barium chloride are combined?
BaSO4 and AgCl will both precipitate from the solution
The ionic compound formed from Ca2+ and PO43- ions has the formula:
Ca3(PO4)2
Which are spectator ions in the redox reaction shown here?
Fe (s) + CuCl2 (aq) → Cu (s) + FeCl2 (aq)
e. Cl- only
How many moles of FeCl3 would be produced if 0.8 moles of iron metal and 0.9 moles of chlorine gas
were combined? Hint: Something might be limiting!
2 Fe (s) + 3 Cl2 (g) → 2 FeCl3 (s)
0.6 mol FeCl3
How many valence electrons are involved in the correctly-drawn Lewis structure of phosphate ion?
32
What are the solubility rules you spoke about in class?
(You won't lose points if you are wrong)

Sulfuric acid has the formula:
a. HSO3
b. HSO4
c. H2SO2
d. H2SO3
e. H2SO
H2SO4
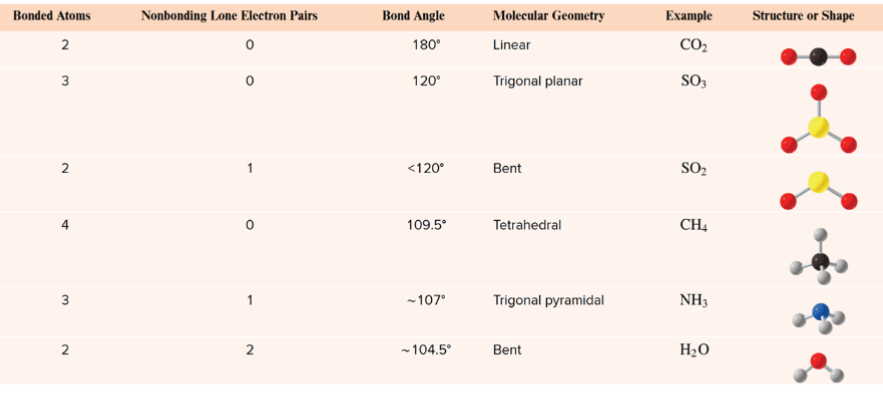
What are all the VSEPR Shapes and the degrees!
(I have the chart from your slide show)