A chemist needs 457.8 g of KMnO4 to make a solution. How many moles of KMnO4 is that?
457.8gKMnO4×(1molKMnO4/158.04gKMnO4)=
2.897 mol KMnO4
Which one of the following classifications is incorrect?(a) H2O(s), molecular solid
(b) C4H10(s), molecular solid
(c) KF(s), ionic solid
(d) SiC(s), covalent solid
(e) S(s), metallic solid
Which one of the following classifications is incorrect?(a) H2O(s), molecular solid
(b) C4H10(s), molecular solid
(c) KF(s), ionic solid
(d) SiC(s), covalent solid
(e) S(s), metallic solid
What are the units expressed for ΔH° rxn?
kJ/mol
Balance the following equation:
Pb(NO3)2 (aq) + NaCl (aq)→NaNO3 (aq) +PbCl2 (s)
Pb(NO3)2 (aq)+2NaCl (aq)→2NaNO3(aq)+PbCl2(s)
How many (sigma bonds/pi bonds) are in a double bond? And how about a single bond?
Can you explain each type of bonding?
double bonds have one σ and one pi (π) bond
single bonds consist of one sigma (σ) bond
sigma (end to end, stronger), pi (side to side, weaker)
Note: as the bond strength increases, the bond length decreases. Thus, we find that triple bonds are stronger and shorter than double bonds between the same two atoms; likewise, double bonds are stronger and shorter than single bonds between the same two atoms.
How many moles of Na2CO3 are in 10.0 mL of a 2.0 M solution?
M = moles of solute / liters of solution
2.0 M = x / 0.0100 L <--- note the conversion of mL to L
x = 0.020 mol
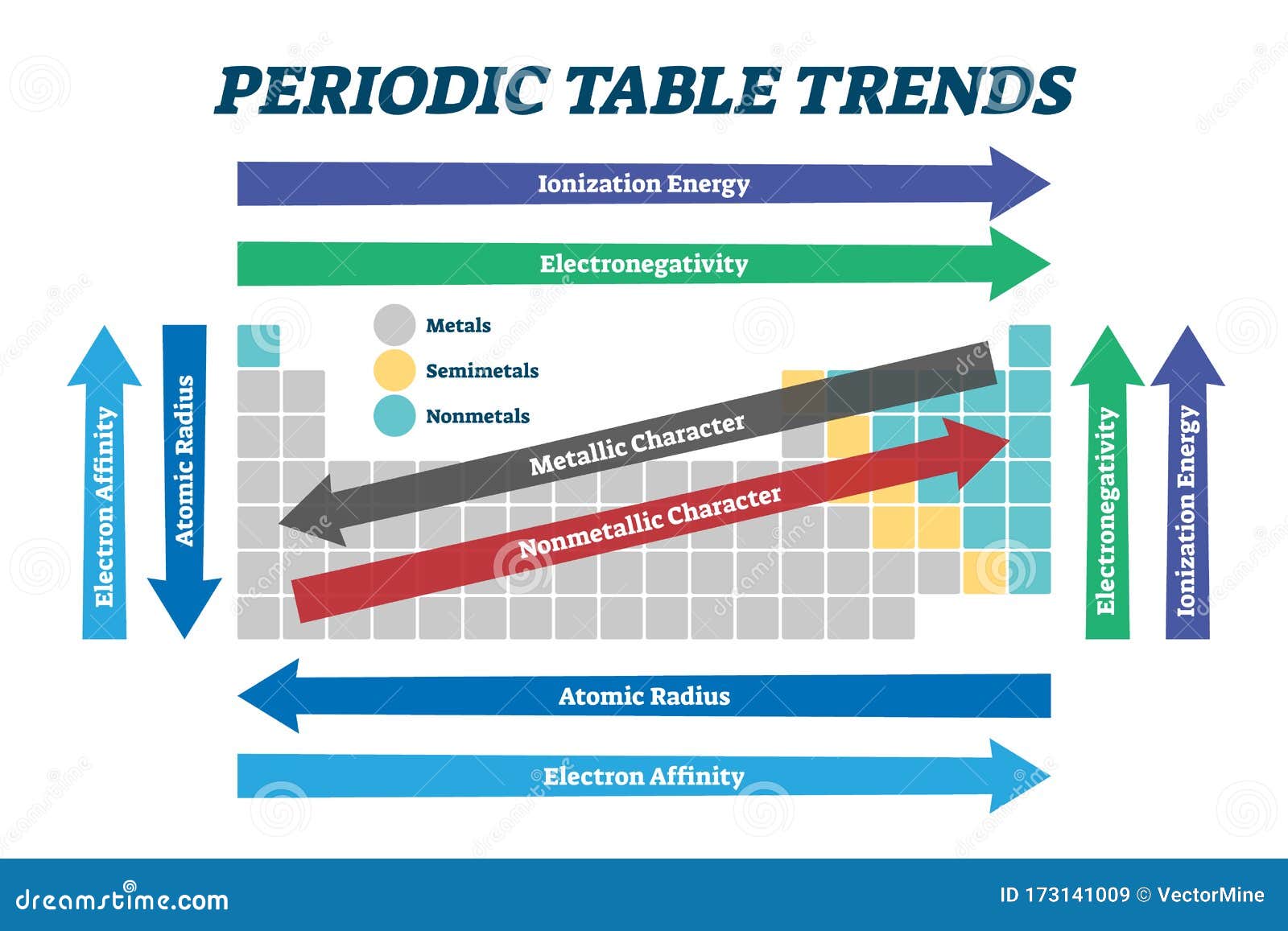
Describe the important periodic trends
What is the most stable form (ΔH°=0) of carbon's allotropes (diamond and graphite)?
graphite
What do the coefficients in the following equation represent?
Na2S + 2AgI → 2NaI + Ag2S
mols, molecules
Assign oxidation numbers to the atoms in the following ion
SO4 2-
SO42-
(+6) + 4(-2) = -2
How many Kelvins are in -15.5 oC
-15.5 oC + 273 = 258K
Knowing that the electron configuration of Al is [Ne] 3s² 3p¹, what is the charge of this ion?
3+
You find that the air trapped in a tube occupies 35.5 cm3 at 2.34 atm. By adding mercury to the tube, you increase the pressure on the trapped air to 3.45 atm. Assuming constant temperature, what is the new volume of air (in L)?
You find that the air trapped in a tube occupies 35.5 cm3 at 2.34 atm. By adding mercury to the tube, you increase the pressure on the trapped air to 3.45 atm. Assuming constant temperature, what is the new volume of air (in L)?
Solved:
P1V1=P2V2
35.5 cm3 x (1 mL/1 cm3) x (1 L/1000 mL)= 0.0355 L
0.0355L(2.34atm)=V2(3.45atm)
Solve for V2
V2=0.0241 L
Write the net ionic equation for 3CuCl2(aq)+2K3PO4(aq)→6KCl(aq)+Cu3(PO4)2(s)
total: 3Cu2+(aq)+6Cl−(aq)+6K+(aq)+2PO3−4(aq)→6K+(aq)+6Cl−(aq)+Cu3(PO4)2(s)
net: 3Cu +2(aq)+2PO4 -3(aq)→Cu3(PO4)2(s)
Describe each inter molecular force from strongest to weakest
ion-dipole, hydrogen bonding, dipole-dipole, and dispersion force
Calculate the number of oxygen atoms and its mass (g) in 50 g of calcium carbonate
calcium carbonate, CaCO3
Molecular mass of CaCO3= 40+12+3×16 = 100
50g(1mol/100g)(3molO/1molCaCO3)(6.022x10^23atoms/1mol) = 9.033×10^23 atoms
Mass of Oxygen atoms = 1.5mol O × 16g/1mol = 24 g
Explain the difference between the copper in the following compounds:
copper (II) sulfate
copper (I) sulfate
Write the equations for each.
Cu+2, Cu+1
copper (II) sulfate CuSO4
copper (I) sulfate Cu2SO4
Calculate ΔH° for the reaction:
Na2O(s) + SO3(g) --> Na2SO4(g) given the following information:
1. Na(s) + H2O(l) --> NaOH(s) + 1/2 H2(g)
ΔH°(1)=-146 kJ
2. Na2SO4(s) + H2O(l) --> 2NaOH(s) + SO3(g)
ΔH°(2)=+418 kJ
3. 2Na2O(s) + 2H2(g) --> 4Na(s) + 2H2O(l)
ΔH°(3)=+259 kJ
(1) x2 = -292
(2) flip = -418
(3) x 1/2 = 129.5
-581 kJ
Group the following into Strong Acids vs Weak Acids:
HF, CH3COOH, H2SO4, H3PO4, HCl
WEAK: HF, CH3COOH (Carboxylic acids are weak), H3PO4
STRONG: H2SO4, HCl
What is the valence bond hybridization of the central atom in each of the following?
(a) BeH2
(b) PO3-4
What is the valence bond hybridization of the central atom in each of the following?
(a) BeH2 linear, sp
(b) PO3-4 tetrahedral, sp3
How many moles of Na are needed to make 4.5 liters of a 1.5 M Na solution?
A solution of molarity 1.5 M, requires 1.5 mol of Na to every liter of solution.
M=mol/L
4.5L requires 6.75 mol of Na, as
4.5(L)x1.5(mol/L)=6.75 (mol)
Which has the lowest boiling point considering molecular size and inter molecular forces?
(a) HF
(b) HCl
(c) HBr
(d) HI
(e) H2SO4
Which has the lowest boiling point?
(a) HF (b) HCl (c) HBr(d) HI(e) H2SO4
Between HCl molecules only small dispersion
HCl < HBr < HI < HF
(If you increase molecular weight, you increase the london dispersion and boiling point as in the order of HCl, HBr, and HI. And HF has the highest boiling point because it has H bonds)
Calculate ΔH° rxn for the following:
CH4 (g) + 2O2 (g) ---> CO2 (g) + 2H2O(l)
and the following values are given:
(ΔH° f CH4= -75 kj/mol, ΔH° f O2= 0 kj/mol, ΔH° f CO2= -394 kj/mol, ΔH° f H2O= -284 kj/mol)
Is this an endothermic or exothermic reaction?
ΔH° rxn = [1 mol xΔH° f (CO2 ) +2 mol x ΔH° f (H2O)]-[1 mol x ΔH° f (CH4)]
−887 kJ/mol exothermic
2 NaCl + Pb(NO3)2 --> 2 NaNO3 + PbCl2
How many grams of lead (II) chloride are produced from the reaction of 15.3 g of NaCl and 60.8 g of Pb(NO3)2? What is the limiting reactant?
LR-->NaCl
36.4 g PbCl2 is produced
bonus points: 17.4 g Pb(NO3)2 will be left over/excess
List every single element in the periodic table that exists diatomically
*hint: remember a certain mnemonic device?
/what-are-the-seven-diatomic-elements-606623-v3-5b562dab46e0fb0037fee8c7.png)