Which temperature scale is necessary for calculating gas law problems?
Kelvin
Write the formula for Boyle's Law
P1V1 = P2V2
Write the formula for Charles' Law
V1/T1 = V2/T2
The amount of space that a gas occupies is known as its ____________________
Volume
What is the formula for converting between Celsius and Kelvin?
Tk = Tc + 273.15
The pressure of a gas and the volume of a gas are _____________________ proportional
Inversely
The volume of a gas and the temperature of a gas are ___________________________ proportional
directly
Write the equation for the combined gas law
P1V1/T1 = P2V2/T2
When a gas exerts force on the side of its container, it is known as _____________________
Pressure
What does STP stand for?
Standard temperature and pressure, 1 atm and 273.15 K
Temperature, amount
Charles' Law only applies as long as the _________________ and ____________________ of a gas remain constant.
Pressure, amount
What is the volume of a gas at 2.00 atm and 200.0 K if its original volume was 300.0 L at 0.250 atm and 400.0 K?
18.8 L
What is the formula for the ideal gas law?
PV=nRT
The following graph represents which gas law?
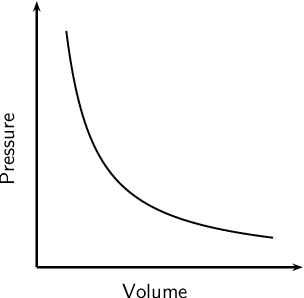
Boyle's Law
I have a balloon that can hold 100 liters of air. If I blow up this balloon with 3 moles of oxygen gas at a pressure of 1 atm, This is the temperature of the balloon.
R= 0.0821 L*atm/mole*K
406 Kelvin or 133 degrees C
The following graph depicts which gas law?
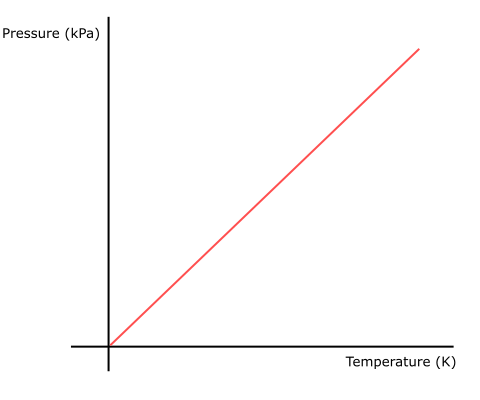
Gay-Lussac's Law
500.0 mL of a gas is collected at 745.0 mm Hg. What will the volume be at standard pressure?
490.1 mL
A gas occupies 1.00 L at a standard temperature. What is the volume at 330.0 oC?
2.22 L
What is the final volume (in Liters) of a 400.0 mL gas sample that is subjected to a temperature change from 22.0oC to 30.0oC and a pressure change from standard pressure to 360 mm Hg?
0.867 L
This is the pressure in a 2120 milliliter tank containing 2.50 mol of argon gas at 25 degree Celsius.
Ideal PV=nRT
28.9 atm