The theory that describes the behavior of gases in term of the motion of gas particles is __________.
the kinetic molecular theory
A weather balloon is released into the atmosphere. You know the initial volume, temperature, and air pressure. What information will you need to predict its volume when it reaches its final altitude? Which law would you use to calculate this volume?
Combined gas law
Find the density (g/L) of a gas at STP with a molar mass of 44.0 g/mol.
1.96 g/L
All of the following are properties of ideal gases except:
a. small amounts of energy are lost during collisions between gas molecules
b. volume occupied by molecules is negligible compared to the volume occupied by the gas
c. gas molecules do not interact with each other except during collisions
d. collisions between gas molecules are completely elastic
a. small amounts of energy are lost during collisions between gas molecules
What is the only state in America whose name ends in "k"?
New York
A particular amount of ideal gas occupies 3.0 L at 27 degrees Celsius. Calculate the decrease in volume observed if the gas is cooled down to 17 degrees Celsius. Report your answer using one significant figure.
0.1 L
The phase diagram below is for mysterious compound X. If you have a bottle of compound X at a pressure of 45 atm and a temperature of 100 degrees Celsius, what phase change will occur when raising the temperature to 400 degrees Celsius?
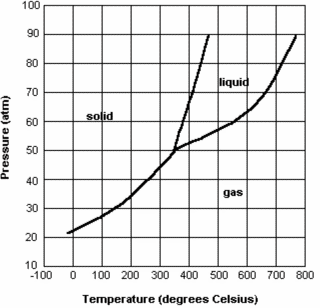
sublimation (solid --> gas)
A sample of O2 gas initially at STP is compressed to a smaller volume at constant temperature. What effect does this change have on
(a) the average kinetic energy of the molecules
(b) their average speed
(c) the number of collisions they make with the container walls per unit time
a) unchanged - because the average kinetic energy of the O2 molecules is determined only by temperature
b) unchanged - because the average kinetic energy of the molecules does not change
c) increases - the molecules are moving in a smaller volume (but with the same speed) so they will encounter the wall more often
At STP, a 0.50 mol sample of hydrogen gas and a separate 1.0 mol sample of oxygen gas have the same
a. average molecular kinetic energy
b. average molecular speed
c. volume
d. chemical formula
a. average molecular kinetic energy
Which element gained notoriety from poisoning the waters around Japan?
Which one of the following statements is not consistent with the kinetic molecular theory of gases?
a. individual gas molecules are relatively far apart
b. the average kinetic energies of different gases are different at the same temperature
c. there is no net gain or loss of the total kinetic energy in collisions between gas molecules
d. the theory explains most of the observed behavior of gases at ordinary temperatures and pressures
b. the average kinetic energies of different gases are different at the same temperature
The phase diagram below is for mysterious compound X. If you wanted to, could you drink compound X? Explain why or why not.

No. We cannot drink a substance at 350 degrees Celsius and 50 atm and these conditions are too extreme for human survival
Which of the following statements is false?
a. The properties of nitrogen gas will deviate more from ideality at -100 degrees Celsius than 100 degrees Celsius
b. Molecules of CH4 gas at high pressures and low temperatures have no attractive forces between each other
c. Molecules of an ideal gas have no significant volume
d. real gases do not always obey the ideal gas law
b. Molecules of CH4 gas at high pressures and low temperatures have no attractive forces between each other
What total gas volume (in liters) at 520 degrees Celsius and 880 torr would result from the decomposition of 33 g of potassium bicarbonate according to the equation: 2KHCO3(s) --> K2CO3(s) + CO2(g) + H2O(g)
1 atm = 760 torr
19 L
Category: Language
The boy: "Tengo un examen de quimica la semana que viene"
What did the boy say?
a. I made an airbag in chemistry class last week.
b. I have a chemistry test next week.
c. I need my calculator but I forgot it at home.
d. I have a periodic table on my desk.
b. I have a chemistry test next week.
Tennis balls are usually filled with N2 gas to a pressure above atmospheric pressure to increase their bounce. If a tennis ball has a volume of 144 cm3 and contains 0.33 g of N2 gas, what is the pressure inside the ball at 24 degrees Celsius?
2.0 atm
Central science textbook page 393
A rigid 5.00 L cylinder contains 24.5 g of N2(g) and 28.0 g of O2(g). Calculate the total pressure, in atm, of the gas mixture in the cylinder at 298 K.
8.56 atm
Which one of the following statements about the following reaction is false?
CH4(g) + 2O2(g) --> CO2(g) + 2H2O(g)
a. Every CH4 molecule that reacts produces two water molecules
b. If 32.0 g of oxygen reacts with excess methane, the maximum amount of carbon dioxide produced will be 22.0 g
c. If 11.2 liters of CH4 react with an excess of oxygen, the volume of carbon dioxide produced at STP is 30.8 liters
c. If 11.2 liters of CH4 react with an excess of oxygen, the volume of carbon dioxide produced at STP is 30.8 liters
Solid potassium chlorate decomposes into solid potassium chloride and oxygen gas. What volume of oxygen, collected at 25.0 degrees Celsius and 101 kPa, can be prepared by decomposition of 37.9 g of potassium chlorate?
11.4 L
What fish is shown below?

a. frog fish
b. bat fish
c. cow fish
d. fish fish
a. frog fish
A neon sign is made of glass tubing whose inside diameter is 2.5 cm and whose length is 5.5 m. If the sign contains neon at a pressure of 1.78 torr at 35 degrees Celsius, how many grams of neon are in the sign? (The volume of a cylinder is πr2h and 1 atm = 760 torr)
5.0 x 10-3 g Ne
In an experiment reported in the scientific literature, male cockroaches were made to run at different speeds on a miniature treadmill while their oxygen consumption was measured. In one hour, the average cockroach running at 0.08 km/hr consumed 0.8 mL of O2 at 1 atm pressure and 24 degrees Celsius per gram of insect mass. How many moles of oxygen would be consumed in 1 hour by a 5.2 gram cockroach moving at this speed?
2 x 10-4 mol O2
Ammonia (NH3) and chlorine gas react to form nitrogen gas and hydrochloric acid (HCl).How many liters of ammonia gas at 244 torr and 35 degrees Celsius must be used to produce 2.3 kg of HCl gas? (1 atm = 760 torr)
1.7 x 103 L NH3
You go on an airplane and take a water bottle. After drinking all the water, you close the water bottle very tightly while the plane is flying. When you land, what happens to water bottle and why?
When we land the bottle will be crushed. At higher altitudes the pressure is low. When we compare the pressure at a plane in higher altitude with the pressure at earth surface, the pressure on the atmosphere is less. When we close the water bottle tightly from plane, the pressure in the bottle is smaller than the pressure on earth surface. When we land, pressure outside the bottle is higher than that of pressure inside the bottle. There is air outside the bottle pushing in at a higher pressure than the air inside the bottle is pushing out. So, the bottle gets crushed.
What is the only US state whose name is just one syllable?
Maine