The electrons in the outer energy level
What are valence electrons?
A part of an atom with a negative charge
What is an electron?
The modern periodic table is organized according to this.
What are by increasing atomic number?
Atomic mass - Atomic number
What is the formula for finding the neutrons of an atom?
Second row, 18th column
What is Neon?
These elements comprise columns 3 - 12 on the periodic table.
WHat are transition metals?
The location of protons and neutrons.
What is inside the nucleus?
The location of electrons.
What is outside the nucleus?
Columns on a periodic table
What are groups?
Arsenic (As) has 33.
What are protons and electrons?
Found in a zigzag line on the periodic table.
What are metalloids?
Except for helium, the elements in this group all have 8 electrons in their outer level.
What is group 18 or noble gases.
The atomic number tells you this.
What is the number of protons (and electrons) an atom of an element has?
The maximum number of electrons the 2nd orbital, or energy shell, can hold.
What is 8.
Rows on the periodic table
What are periods?
Arsenic (As) has 42.
What are neutrons?
An atom with a completely full outer most (valence) energy level is this.
What is nonreactive or noble gases?
THese elements, found at the bottom of the periodic table, are elements 89-103.
What are the actinides?
The maximum number of electrons the 1st orbital, or energy shell, can hold.
What is 2?
A part of an atom with a positive charge
What is a proton?
The majority of the elements on the periodic table are this.
What are metals?
These are drawings that represent where the parts of an atom are located.
What are atomic models.
Tiny particles that make up all matter.
What are atoms?
Fourth period noble gas.
What is krypton?
This model looks like this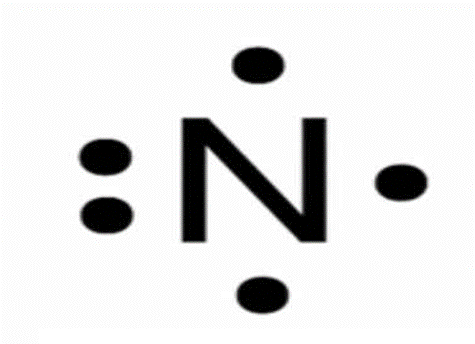
What is Lewis dot model.
A subatomic particle with no charge.
What is a neutron?
Elements in the same group share this.
What are the same number of valence electrons
Elements in group 1 have only 1 valence electron and are considered this.
What are elements that alkali metals?
Located on the right side of the zigzag line.
What are nonmetals?
THese elements, also found at the bottom of the periodic table, are elements 57-71.
What are lanthanides?
The number of electrons that can go into the third level.
What is 18?
THis particle has practically no mass.
What is an electron?
Fourth period halogen
What is Bromine?
Its atomic symbol is Fe
What is iron?
Third period Alkaline earth metal.
What is magnesium?
These elements are malleable, ductile, and have a metallic luster.
What are metals?