A _______ is a state of matter with both a fixed shape and a fixed volume.
What is solid
A pure substance is made out of ______ types of particles
What is one
All particles are in constant ________
What is motion
This image is likely particles in a...
What is the term for a substance that dissolves in a solvent to form a solution?
What is solute?
A _______ is a state of matter with no fixed shape, but a fixed volume.
What is liquid
A mixture is made up of _____ kinds of particles
What is many?
All matter is made up of ________
What is particles
 These particles are likely those in a....
These particles are likely those in a....
What is Mechanical mixture
What do you call the maximum amount of a solute that can dissolve in a given amount of solvent at a specific temperature?
What is solubility?
A _______ is a state of matter with no fixed shape or volume.
What is gas
This image is likely particles in a
what is pure substance
What is temperature
A granola bar is a...
Name one factor that can affect the solubility of a substance.
What is temperature, pressure, or the nature of the solute and solvent?
This image likely shows particles in a.... 
These particles are likely in a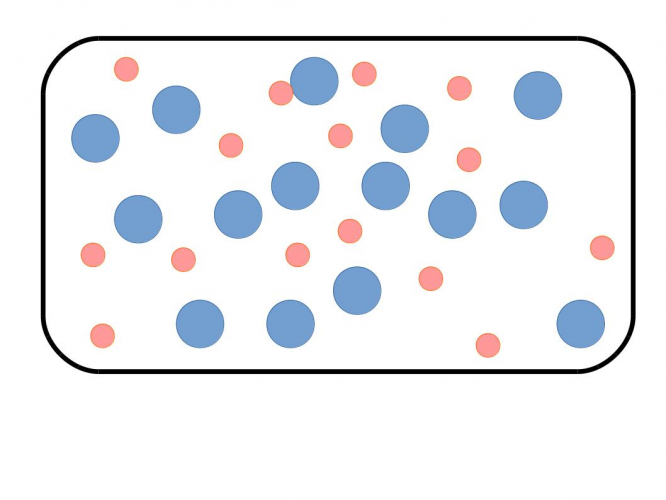
Particles ______ one another so they don't fly apart
Ketchup is an example of a...
What is solution
What term describes how much solute is present compared to the amount of solvent or solution?
What is concentration?
This image likely shows particles in a...
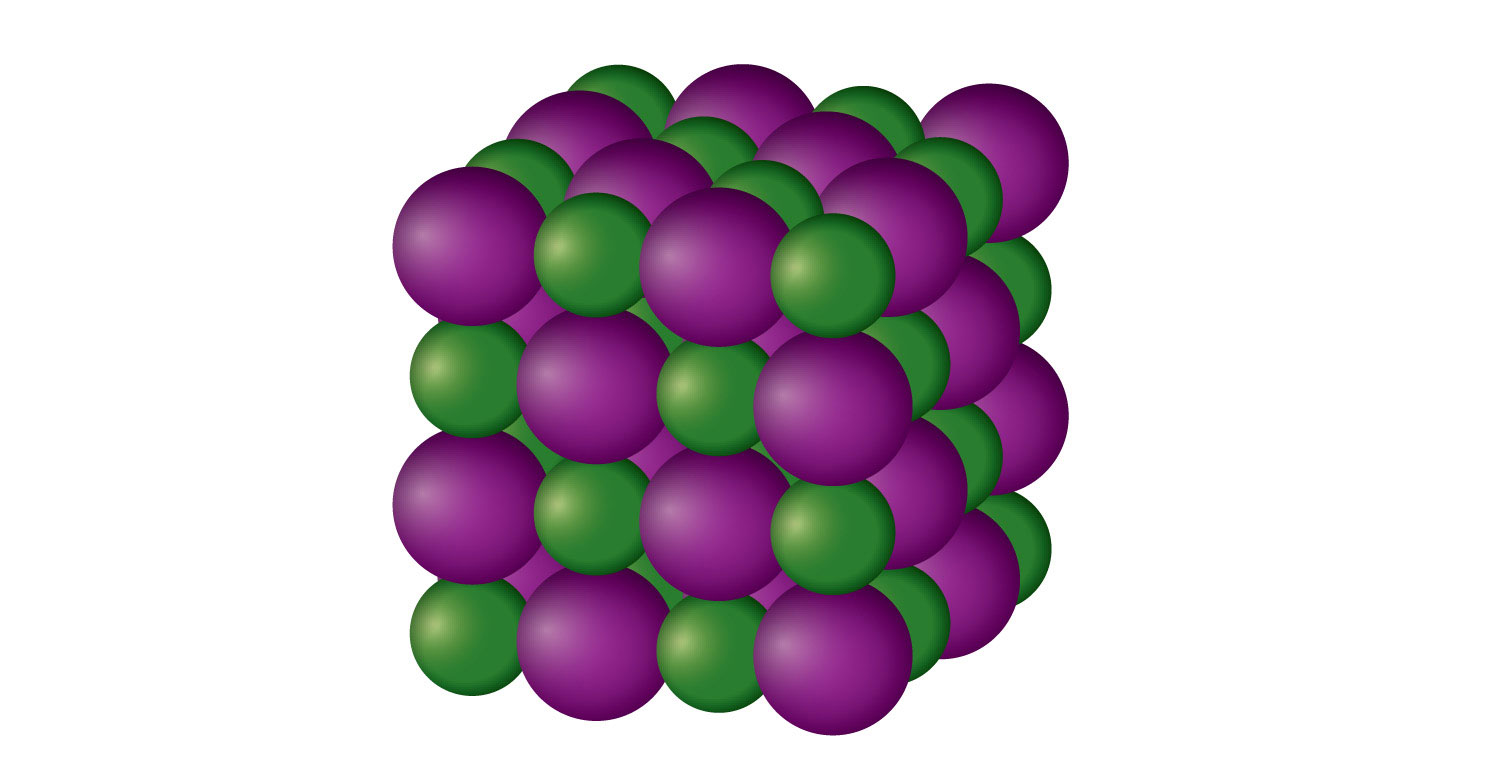
What is solid
Water and oil is an example of a
What is mixture
Even though we can't see them, there are tiny ______ between the particles
What are spaces
What term is used to describe a solution in which no more solute can be dissolved at a given temperature and pressure, and any additional solute will remain undissolved?
What is a saturated solution?