Cysteine is the only natural amino acid to contain this functional group.
Thiol/Mercaptan
The cutoff value in nanometers between UV and visible radiation
400 +/- 50
Most common stereochemistry of the alpha carbon in amino acids.
L
The scientist who proposed energy quantization as a resolution to the ultraviolet catastrophe.
Max Planck
Number of hydrochloric acid molecules in 1 L of 5.0 M HCl.
10^24
The oxygen containing functional group in triglycerides.
Esters
The component of a spectrometer is pictured here

Monochromator
Number of methylene carbons in aspartic acid.
1
The sign of entropy change in vaporization.
Positive
Energy in joules of 790 nm light.
10^-19
The group on serine and threonine that is frequently the substrate of protein kinase activity.
Hydroxyl/Alcohol
The multiplicity of the terminal methyl peak in an ethyl formate NMR spectrum.
3/triplet
The charge on peptide EAVYPAAKCRLI at pH 7.
+1
The expression for the momentum operator.
-iħ (d/dx)
Force constant of a typical -OH bond in N/m.
10 ^ 2
The order of the bonds connecting the nitrogens of an azide group.
2
The typical units of collisional cross section (as tabulated in mobcal).
Å 2
The typical barrier of cis/trans isomerization in peptides.
20 kcal/mol
How the time-evolution of a first-order decompostion reaction goes.
Exponential decay
Number of publications in JACS.
10 ^ 5
The functional group pictured here: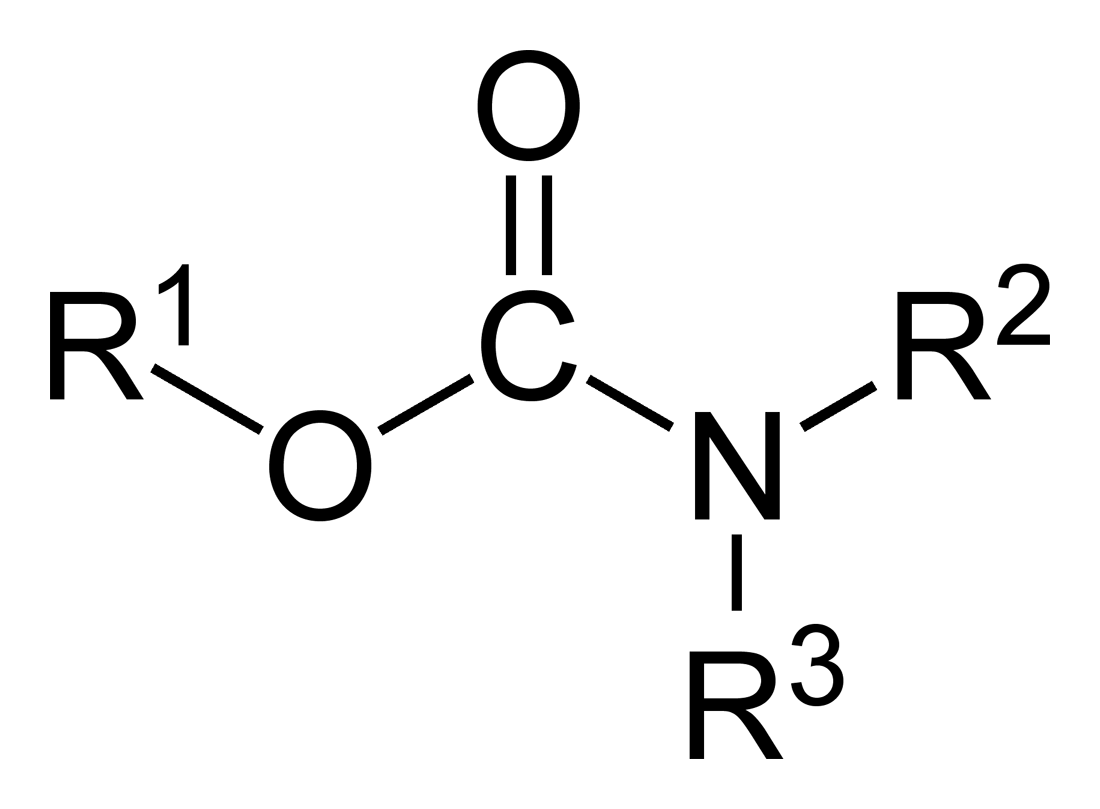
Carbamide
The equation governing the mass filtering of a quadrupole.
Mathieu equation
Draw the structure of O-phosphorylated serine.

The expectation value of x in a particle in a box wavefunction with length L.
L/2
The vibrational partition function of CO2 at RT.
10 ^ 0